A) universal law
B) first law of thermodynamics
C) law of mass conservation
D) law of conservation of energy
E) law of constant energy
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When 0.7521 g of benzoic acid was burned in a calorimeter containing 1000.g of water,a temperature rise of 3.60°C was observed.What is the heat capacity of the bomb calorimeter,excluding the water? The heat of combustion of benzoic acid is -26.42 kJ/g.
A) 15.9 kJ/°C
B) 4.2 kJ/°C
C) 5.5 kJ/°C
D) 1.3 kJ/°C
E) 752 kJ/°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system absorbs 21.6 kJ of heat while performing 6.9 kJ of work on the surroundings.If the initial internal energy,U,is 61.2 kJ,what is the final value of U?
A) 32.7 kJ
B) 46.5 kJ
C) 61.2 kJ
D) 75.9 kJ
E) 89.7 kJ
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Your favorite candy bar,Gummy Beakers,contains 1.2 × 106 J of energy while your favorite soft drink,Bolt,contains 6.7 × 105 J.If you eat two packs of Gummy Beakers a day and drink 3 cans of Bolt,what percent of your 2000 Calorie daily food intake is left for broccoli,beans,beef,etc.? (1 cal = 4.184 J)
A) 53%
B) 47%
C) 27%
D) 11%
E) 0%
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Chemical reactions in a bomb calorimeter occur at constant pressure.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is not a state function?
A) internal energy
B) volume
C) work
D) pressure
E) enthalpy
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which of the substances below is ΔHof = 0?
A) O2(g)
B) N2(l)
C) Na(g)
D) Xe(l)
E) A and B
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? SiO2(s) + 4HCl(g) → SiCl4(g) + 2H2O(g) Substance ΔH°f(kJ/mol) SiO2(s) -859.3 SiCl4(g) -662.8 HCl(g) -92.3 H2O(g) -241.8
A) -1856.2 kJ/mol
B) -1372.6 kJ/mol
C) -47.0 kJ/mol
D) 82.1 kJ/mol
E) 530.6 kJ/mol
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If two solutions are mixed together in a container and the container "feels hot",then
A) the reaction is endothermic.
B) the reaction is exothermic.
C) the energy of the universe is increased.
D) the energy of both the system and the surroundings is decreased.
E) the energy of the system is increased.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A glass containing 200.0 g of H2O at 20.0°C was placed in a refrigerator.The water loses 11.7 kJ as it cools to a constant temperature.What is its new temperature? The specific heat of water is 4.184 J/g·°C.
A) 0.0°C
B) 4.0°C
C) 6.0°C
D) 14.0°C
E) 34.0°C
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two solutions (the system) ,each with a volume of 25.0 mL at 25.0°C,are mixed in a beaker.A reaction occurs between them,and the temperature rises to 35.0°C.After the products have equilibrated with the surroundings,the temperature is again 25.0°C and the total volume is 50.0 mL.No gases are involved in the reaction.Which relationship concerning the change from initial to final states (both at 25.0°C) is correct?
A) ΔU = 0
B) ΔH = 0
C) ΔU > 0
D) q = 0
E) W = 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following two representations of chemical reactions.Each sphere represents 1 mol of atoms. 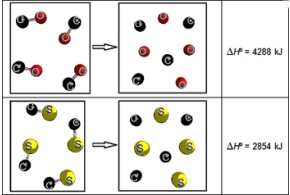 What is ΔHºrxn for the following reaction? CS(g) + O(g) → CO(g) + S(g)
What is ΔHºrxn for the following reaction? CS(g) + O(g) → CO(g) + S(g)
A) -359 kJ/mol
B) 1434 kJ/mol
C) -7142 kJ/mol
D) -1434 kJ/mol
E) 7142 kJ/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For which reaction is ΔH approximately (or exactly) equal to ΔU?
A) H2(g) + Br2(g) → 2HBr(g)
B) H2O(l) → H2O(g)
C) CaCO3(s) → CaO(s) + CO2(g)
D) 2H(g) + O(g) → H2O(l)
E) CH4(g) + 2O2(g) → CO2(g) + 2H2O(l)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An endothermic reaction causes the surroundings to
A) warm up.
B) become acidic.
C) condense.
D) decrease in temperature.
E) release CO2.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The dissolution of barium hydroxide in water is an exothermic process.Which statement is correct?
A) The enthalpy of solid barium hydroxide plus pure water is less than that of the solution,at the same temperature.
B) The enthalpy of solid barium hydroxide plus pure water is greater than that of the solution,at the same temperature.
C) The enthalpy of solid barium hydroxide plus pure water is the same as that of the solution,at the same temperature.
D) The temperature of the solution is lower than of the barium hydroxide and water before mixing.
E) When barium hydroxide dissolves in water,the system does work on the surroundings.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The combustion of pentane produces heat according to the following thermochemical equation. C5H12(l) + 8O2(g) → 5CO2(g) + 6H2O(l) ΔH°rxn = -3510 kJ/mol How many grams of CO2 is produced per 2.50 × 103 kJ of heat released?
A) 6.27 g
B) 31.3 g
C) 61.8 g
D) 157 g
E) 220 g
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system delivers 1275 J of heat while the surroundings perform 854 J of work on it.What is ΔU of the system?
A) -2129 J
B) -421 J
C) +421 J
D) +2129 J
E) -1275 J
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A student used a bomb calorimeter to determine the heat of combustion of an unknown compound.If the student ignored the heat capacity of the bomb calorimeter,the heat capacity of the unknown compound would be _________ the accepted value.
A) greater than (i.e.,less negative)
B) less than (i.e.,more negative)
C) equal to
E) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much heat is released if 7.15 g CaO(s) is added to 152 g of H2O(l) ? CaO(s) + H2O(l) → Ca(OH) 2(s) ΔH°rxn = -64.8 kJ/mol
A) 7.68 kJ
B) 8.26 kJ
C) 508 kJ
D) 547 kJ
E) 555 kJ
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? NO2(g) + CO(g) → CO2(g) + NO(g) Substance ΔH°f(kJ/mol) NO(g) +90.4 NO2(g) +33.85 CO(g) -110.5 CO2(g) -393.5
A) 339.6 kJ
B) 379.8 kJ
C) -226.5 kJ
D) -339.6 kJ
E) -379.8 kJ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 134
Related Exams