A) The reactants will be favored because ammonia is a stronger base than the sulfate anion.
B) The products will be favored because the hydrogen sulfate ion is a stronger acid than the ammonium ion.
C) Neither reactants nor products will be favored because all of the species are weak acids or bases.
D) The initial concentrations of the hydrogen sulfate ion and ammonia must be known before any prediction can be made.
E) This reaction is impossible to predict,since the strong acid and the weak base appear on the same side of the equation.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which represents a weak base B with Kb= 2 ×10-3? (Each circle represents 1.0 ×10-3 mol of atoms,and the volume of each box is 1.0 L.Solvent water molecules are not shown for clarity.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Weak acids have weak conjugate bases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the weak bases below and their Kb values: C6H7O- Kb = 1.3 × 10-10 C2H5NH2 Kb = 5.6 × 10-4 C5H5N Kb = 1.7 ×10-9 Arrange the conjugate acids of these weak bases in order of increasing acid strength.
A) C5H5NH+< C6H7OH < C2H5NH
B) C6H7OH < C5H5NH+ < C2H5NH
C) C5H5NH+< C2H5NH3+ < C6H7OH
D) C6H7OH < C2H5NH3+< C5H5NH+
E) C2H5NH3+< C5H5NH+ < C6H7OH
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which aqueous solution has the highest pH?
A) 0.1 M CH3COOH,pKa = 4.7
B) 0.1 M CuCl2,pKa = 7.5
C) 0.1 M H3C6H5O7,pKa = 3.1
D) 0.1 M ZnCl2,pKa = 9.0
E) pure water
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the conjugate acid of the acetate ion?
A) CH3O2-
B) COOH
C) HC2H3O2
D) H2O
E) CH4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a strong acid?
A) H2CO3
B) H2SO3
C) H2SO4
D) H3PO4
E) CH3COOH
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a strong acid?
A) CH3COOH
B) H2SO3
C) NH3
D) H3PO4
E) HClO3
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Kw = 1.0 × 10-14 under all conditions.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
Amphoteric oxides are compounds that exhibit both acidic and basic behavior.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the reaction CaO(s) + SO2(g)  CaSO3(s) ,
CaSO3(s) ,
A) O2- acts as a Lewis base,and SO2 acts as a Lewis acid.
B) Ca2+ acts as a Lewis base,and SO42- acts as a Lewis acid.
C) SO32- acts as a Lewis base,and SO2 acts as a Lewis acid.
D) SO2 acts as a Lewis base,and O2- acts as a Lewis acid.
E) SO2 acts as a Lewis base,and Ca2+ acts as a Lewis acid.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution is prepared by adding 0.10 mol of sodium fluoride,NaF,to 1.00 L of water.Which statement about the solution is correct?
A) The solution is basic.
B) The solution is neutral.
C) The solution is acidic.
D) The concentrations of fluoride ions and sodium ions are equal.
E) The concentrations of hydronium ions and hydroxide ions are equal.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Find the pH of a 0.20 M aqueous solution of dichloroacetic acid,for which Ka = 3.32 × 10-2.
A) 0.75
B) 2.71
C) 1.05
D) 2.35
E) 1.18
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the strongest acid?
A) HBrO3
B) HFO3
C) HIO3
D) HClO3
E) All are of equal strength.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a representation of an aqueous solution of a weak acid HA at equilibrium.(Each circle represents 1.0 ×10-3 mol of atoms,and the volume of the box is 1.0 L.Solvent water molecules are not shown for clarity.) 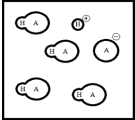 What is Ka of HA?
What is Ka of HA?
A) 1.0 ×10-6
B) 6.0 ×10-3
C) 2.5 ×10-4
D) 3.6 ×10-5
E) 1.0 ×10-9
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hard water deposits (calcium carbonate) have built up around your bathroom sink.Which of these substances would be most effective in dissolving the deposits?
A) ammonia
B) bleach (sodium hypochlorite)
C) lye (sodium hydroxide)
D) vinegar (acetic acid)
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The substance (CH3CH2) 2NH is considered to be
A) a weak acid.
B) a weak base.
C) a strong acid.
D) a strong base.
E) a neutral compound.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is [OH-] for a solution at 25°C that has [H3O+] = 2.35 × 10-3 M?
A) 4.26 × 10-5 M
B) 2.35 ×10-11 M
C) 4.26 × 10-12 M
D) 2.35 × 10-17 M
E) 2.35 ×10-3 M
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is a Lewis acid but not a Brønsted acid?
A) Fe3+
B) H3O+
C) HSO4-
D) NH3
E) H2O
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a 0.050 M LiOH solution?
A) 1.30
B) 3.00
C) 11.00
D) 12.39
E) 12.70
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 133
Related Exams