Filters
Question type
A) 11.08
B) 10.74
C) 10.81
D) 10.55
E) 10.49
F) B) and E)
G) A) and D)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Question 72
Multiple Choice
Calculate the pH at the equivalence point for the titration of 50.0 mL of 0.20 M NH3 with 0.20 M HCl. [Kb(NH3) = 1.8 × 10-5]
A) 2.87
B) 4.98
C) 5.13
D) 7.00
E) 11.13
F) C) and E)
G) All of the above
G) All of the above
Correct Answer

verified
Correct Answer
verified
Question 73
True/False
Increasing the concentrations of the components of a buffer solution will increase the buffer capacity.
A) True
B) False
B) False
Correct Answer

verified
Correct Answer
verified
Question 74
Multiple Choice
Which one of the following is the best representation of the titration curve which will be obtained in the titration of a weak diprotic acid H2A (0.10 M) with a strong base of the same concentration?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
F) A) and E)
G) A) and D)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Question 75
Multiple Choice
Below is a representation of the sparingly soluble salt MX at equilibrium in aqueous solution.(Each circle represents 1.0 ×10-3 mol of atoms,and the volume of the box is 1.0 L.Solvent water molecules are omitted for clarity.) 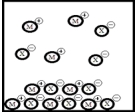 What is Ksp for MX?
What is Ksp for MX?
A) 1.5 ×10-5
B) 6.0 ×10-6
C) 3.6 ×10-1
D) 1.8 ×10-3
E) 9.0 ×10-6
F) C) and D)
G) A) and C)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Question 76
Multiple Choice
Write the ion product expression for calcium phosphate,Ca3(P O4) 2.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) (3 × [Ca2+]) (2 × [P O43- ]
F) A) and C)
G) D) and E)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Question 77
Multiple Choice
Which is more soluble in a basic solution than in pure water?
A) AgF
B) AgCN
C) CO2
D) NaCl
E) CH3COOK
F) All of the above
G) A) and D)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Question 78
Multiple Choice
Calculate the pH of a buffer solution prepared by dissolving 0.20 mole of sodium cyanate (NaCNO) and 1.0 mole of cyanic acid (HCNO) in enough water to make 1.0 liter of solution. [Ka(HCNO) = 2.0 × 10-4]
A) 0.00
B) 3.00
C) 3.70
D) 4.40
E) 5.00
F) B) and D)
G) A) and C)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Question 79
Short Answer
What name is given to a solution that contains a weak acid and its conjugate base?
Correct Answer

verified
Correct Answer
verified
Showing 121 - 129 of 129
Related Exams