A) 90°
B) 109.5°
C) 120°
D) 180°
F) All of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Avobenzone is an active ingredient in some common sunscreens. Which of the following is the correct molecular formula for avobenzone? 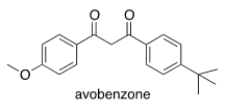
A) C22O22O3
B) C20H22O3
C) C21H23O3
D) C20H24O3
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about electronegativity and the periodic table is true?
A) Electronegativity decreases across a row of the periodic table.
B) Electronegativity increases down a column of the periodic table.
C) Electronegativity increases across a row of the periodic table.
D) Electronegativity does not change down a column of the periodic table.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has nonpolar covalent bonds?
A) HCl
B) N2
C) CHCl3
D) NO
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the order of decreasing bond length for a C-C bond comprised of the following molecular orbitals?
A) I > III > II
B) I > II > III
C) III > II > I
D) II > III > I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the C-H bonding molecular orbitals of ethane, CH3CH3?
A) Csp2 + H1s
B) Csp3 + H1s
C) C2p + H1s
D) Csp + H1s
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about constitutional isomers is true?
A) Constitutional isomers are different molecules having different molecular formula.
B) Constitutional isomers are different molecules having same molecular formula.
C) Constitutional isomers are same molecules having different molecular formula.
D) Constitutional isomers are same molecules having the same molecular formula.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the carbon atom in the methyl cation, (CH3+) ?
A) sp3
B) sp2
C) sp
D) p
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a magnesium cation (Mg2+) ?
A) 1s2, 2s2, 2p6
B) 1s2, 2s2, 2p6, 3s1
C) 1s2, 2s2, 2p6, 3s2
D) 1s2, 2s2, 2p6, 3s2, 3p2
F) B) and C)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following compounds has an atom with an unfilled valence shell of electrons?
A) H2O
B) BCl3
C) CH4
D) CO2
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following ions does carbon have a formal charge? I II III
A) I
B) II
C) III
D) None of the above
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate C-C-C bond angle in propene, CH3CH=CH2?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many different isomers are there for a compound having the molecular formula C3H6O?
A) 4
B) 5
C) 6
D) 7
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds has the largest dipole moment?
A) C-H
B) C-C
C) C-O
D) H-F
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the C-H bonding molecular orbitals of ethylene, H2C=CH2?
A) C2p + H1s
B) Csp + H1s
C) Csp3 + H1s
D) Csp2 + H1s
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following ions does carbon have a formal charge? I II III
A) I
B) II
C) III
D) None of the above
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular C3H8O?
A) 1
B) 2
C) 3
D) 4
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pair does not represent resonance structures? 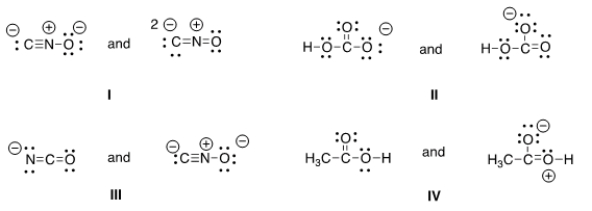
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization for each of the indicated atoms in the following compound? 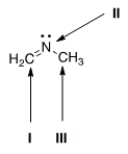
A) I = sp2; II = sp2; III = sp2.
B) I = sp2; II = sp3; III = sp3.
C) I = sp; II = sp2; III = sp3.
D) I = sp2; II = sp2; III = sp3.
F) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Follow the curved arrows to draw the second resonance structure for the ion below. 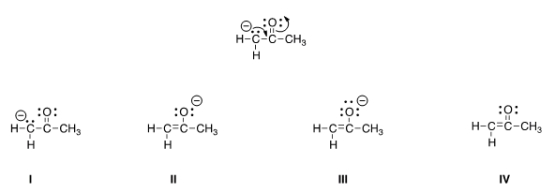
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 70
Related Exams