A) phenylalanine
B) glycine
C) valine
D) alanine
F) A) and B)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Why does the formation of salts of a racemic mixture of N-acetyl valine with a single enantiomer of -methylbenzylamine enable the separation of the two enantiomers of N-acetyl valine?
A) They form amides.
B) They form diastereomers.
C) They form salts.
D) They form meso compounds.
F) A) and C)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following is the correct structure for aspartic acid at pH = 1? 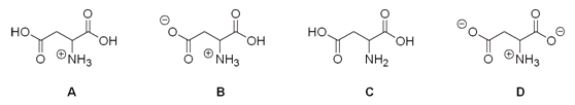
A) A
B) B
C) C
D) D
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is (are) globular protein(s) ?
A) hemoglobin
B) keratins
C) collagens
D) elastins
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes a protein?
A) a dipeptide; two amino acids joined together by one amide bond
B) a tripeptide; three amino acids joined together by two amide bonds
C) a polymer of more than 40 amino acids joined together by amide bonds
D) a polypeptide of any number of amino acids joined together by amide bonds
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the basic steps in the Merrifield peptide synthesis?
A) coupling and hydrolysis
B) hydrolysis and deprotection
C) deprotection and hydrogenation
D) coupling and deprotection
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes the tertiary structure of a peptide?
A) the three-dimensional conformations of localized regions of a protein
B) the particular sequence of amino acids that are joined together by peptide bonds
C) the structure is defined by the -pleated sheet form
D) the three-dimensional shape adopted by the entire peptide
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the IUPAC or common name for the following compound? 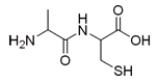
A) cysteinylalanine
B) alanylcysteine
C) serinylalanine
D) alanylserine
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Reaction of a polypeptide, composed of 12 amino acids, with carboxypeptidase A releases Met (C-terminal amino acid) . The polypeptide undergoes partial hydrolysis to give 12 groups of peptides. Use the groups of overlapping amino acids to determine the proper sequence of this polypeptide. Note: Since these lists of peptides are separated by commas, they are not necessarily in the proper sequence. 1. Ser, Lys, Trp 2. Gly, His, Ala 3. Glu, Val, Ser 4. Leu, Glu, Ser 5. Met, Ala, Gly 6. Ser, Lys, Val 7. Glu, His 8. Leu, Lys, Trp 9. Lys, Ser 10. Glu, His, Val 11. Trp, Leu, Glu 12. Ala, Met
A) Met-Ala-Gly-His-Glu-Val-Ser-Lys-Trp-Leu-Glu-Ser
B) Met-Ala-Gly-Glu-His-Ser-Val-Lys-Trp-Leu-Glu-Ser
C) Ser-Glu-Leu-Trp-Lys-Ser-Val-Glu-His-Gly-Ala-Met
D) Ser-Lys-Leu-Trp-Lys-Ser-Val-His-Glu-Gly-Ala-Met
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following correctly describes a peptide bond?
A) a bond between sp3-hybridized atoms only
B) a bond usually found in the s-cis conformation
C) an amide bond
D) the result of the formation of the carbonyl group of one amino acid with a R-group from the other amino acid
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct structure for the amino acid valine? 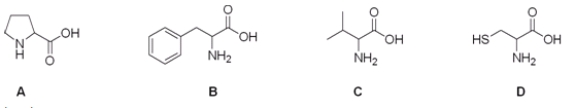
A) A
B) B
C) C
D) D
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the major advantage of the Merrifield synthesis of peptides?
A) It allows for the formation of absolutely no by-products.
B) Impurities can be easily washed away.
C) Excess reagent can be used.
D) It prevents hydrogenation.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is a proposed method for resolving a racemic mixture of (R) - and (S) -phenylalanine. Upon separation of the products, will this method produce (R) - or (S) -phenylalanine? 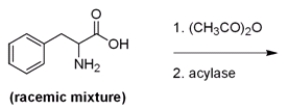
A) (R) -Phenylalanine is produced since the acylase only hydrolyzes the amides of L-amino acids.
B) (S) -Phenylalanine is produced since the acylase only hydrolyzes the amides of L-amino acids.
C) (R) -Phenylalanine is produced since the acylase only hydrolyzes the amides of D-amino acids.
D) (S) -Phenylalanine is produced since the acylase only hydrolyzes the amides of D-amino acids.
F) All of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Below is a 2D chromatogram that shows the separation of five amino acids. In this technique, the amino acid mixture is first separated by chromatography using a polar solvent system. Then the plate is rotated 90°, and the amino acids are further separated by electrophoresis. Identify the spots obtained from a mixture of Trp, Glu, Lys, Ile and Thr. A table with isoelectric points is included to aid in solving this problem. 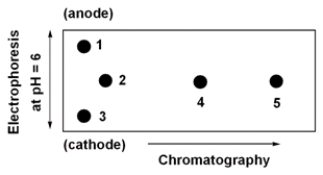 amino acid pI value
Trp
5) 9
Glu
3) 2
Lys
9) 7
Ile
6) 0
Thr
5) 6
amino acid pI value
Trp
5) 9
Glu
3) 2
Lys
9) 7
Ile
6) 0
Thr
5) 6
A) 1=Lys, 2=Thr, 3=Glu, 4=Trp, 5=Ile
B) 1=Glu, 2=Thr, 3=Lys, 4=Trp, 5=Ile
C) 1=Lys, 2=Ile, 3=Glu, 4=Trp, 5=Thr
D) 1=Glu, 2=Ile, 3=Lys, 4=Trp, 5=Thr
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the appropriate sequence of reaction conditions used to synthesize valine from acetamidomalonic ester? 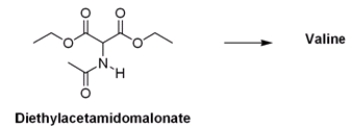
A) (1) NaOH; (2) (CH3) 2CHBr; (3) Cl2, H2O, heat
B) (1) NaOCH2CH3; (2) (CH3) 2CH2CHBr; (3) HCl, H2O, heat
C) (1) NaOCH2CH3; (2) (CH3) 2CHBr; (3) HCl, H2O, heat
D) (1) NaOH; (2) CH3CH2CH2Br; (3) HCl, H2O, heat
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the missing reagent(s) in the reaction below? 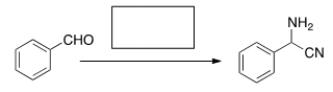
A) NH3
B) HCN
C) NaCN, NH4Cl
D) NaNH2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why can glycine not be resolved into two enantiomers?
A) It has no stereogenic centers.
B) It has no nitrogen atoms.
C) It has no aromatic rings.
D) It cannot be converted into a salt.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the configuration of naturally occurring amino acids?
A) D
B) S
C) L
D) R
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What amino acid is at the N-terminus of the following peptide? 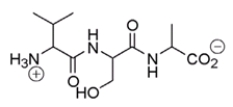
A) valine
B) serine
C) alanine
D) proline
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why is the s-trans form of an amide bond more stable?
A) There is no s-cis form in an amide bond.
B) The oxygen is hydrogen-bonded with the nitrogen atom.
C) The nitrogen is able to be sp2-hybridized.
D) The more sterically bulky groups are farther apart.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 41
Related Exams