A) 0.1 M NaI
B) 0.2 M CuSO4
C) 0.5 M KBr
D) 2.0 M CuCl2
E) 2.0 M CsCl
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
If the density of a solution is less than 1.0 g/mL, its molarity will be greater than its molality.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
A 0.137 m solution of sodium ferrocyanide (Na4Fe(CN)6.10H2O) in water has a freezing point depression of 0.755°C. Calculate the van't Hoff factor (i) under these conditions. (Kf = 1.86°C/m)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Soda drinks bubble when the bottle is opened because
A) the temperature of the soda increases.
B) exposure to atmospheric pressure squeezes the carbon dioxide from solution.
C) the partial pressure of carbon dioxide above the solution is reduced.
D) atmospheric nitrogen molecules displace carbon dioxide molecules.
E) the Henry's law constant changes due to the change in pressure.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Predict the ideal van't Hoff factor (i) for each of the following solutes: a. CaCl2 b. Fe2(SO4)3 c. NH4Cl
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following pairs of ions is arranged so that the ion with the smaller charge density is listed first?
A) K+, Rb+
B) Cl-, K+
C) Cl-, Br-
D) Ca2+, Ba2+
E) None of the above pairs are arranged in this way.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Aqueous ammonia is commercially available in a solution that is 28% (w/w) ammonia. What is the mole fraction of ammonia in such a solution?
A) 0.017
B) 0.023
C) 0.012
D) 0.24
E) 0.29
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
From the following list of aqueous solutions and water, select the one with the highest boiling point.
A) 1.0 m KNO3
B) 0.75 m NaCl
C) 0.75 m CuCl2
D) 2.0 m C12H22O11 (sucrose)
E) pure water
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium fluoride is used for frosting glass. Calculate the molarity of a solution prepared by dissolving 78.6 g of KF in enough water to produce 225 mL of solution.
A) 0.304 M
B) 0.349 M
C) 1.35 M
D) 3.29 M
E) 6.01 M
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Octane and nonane are liquids which are components of gasoline. Their vapor pressures at 25°C are 13.9 torr and 4.7 torr, respectively. What is the vapor pressure of a mixture consisting of 1 mole of each of these compounds?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of ethanol (C2H6O) in water is sometimes used as a disinfectant. 1.00 L of this solution contains 553 g of ethanol and 335 g of water. What is the mole fraction of ethanol in this solution?
A) 0.377
B) 0.392
C) 0.553
D) 0.608
E) 0.623
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Isoamyl salicylate ( 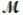 = 208.25 g/mol) has a pleasant aroma and is used in perfumes and soaps. Which of the following combinations gives a 0.75 m solution of isoamyl salicylate in ethyl alcohol (d = 0.7893 g/mL) ?
= 208.25 g/mol) has a pleasant aroma and is used in perfumes and soaps. Which of the following combinations gives a 0.75 m solution of isoamyl salicylate in ethyl alcohol (d = 0.7893 g/mL) ?
A) 117.2 g isoamyl salicylate in 950.0 mL of ethyl alcohol
B) 117.2 g isoamyl salicylate in 750.0 mL of ethyl alcohol
C) 117.2 g isoamyl salicylate in 750.0 mL of solution
D) 117.2 g isoamyl salicylate in 592.0 g of ethyl alcohol
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A solution of sucrose (sugar) in water is in equilibrium with solid sucrose. If more solid sucrose is now added, with stirring,
A) the concentration of the solution will increase.
B) the concentration of the solution will decrease.
C) the concentration of the solution will remain the same.
D) the volume of solution will increase.
E) a supersaturated solution will be produced.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Potassium hydrogen phosphate is used in the preparation of non-dairy powdered creamers. Calculate the molarity of a solution prepared by dissolving 238 g of K2HPO4 in enough water to produce 275 mL of solution.
A) 0.732 M
B) 0.865 M
C) 2.66 M
D) 4.97 M
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Children under the age of six with more than 0.10 ppm of lead in their blood can suffer a reduction in I.Q. or have behavior problems. What is the molality of a solution which contains 0.10 ppm of lead?
A) 4.8 × 10-10 m
B) 4.8 × 10-7 m
C) 4.8 × 10-4 m
D) 4.8 × 10-1 m
E) none of the above
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Barbiturates are synthetic drugs used as sedatives and hypnotics. Barbital ( 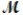 = 184.2 g/mol) is one of the simplest of these drugs. What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid? Kb = 3.07°C/m; boiling point of pure acetic acid = 117.9°C
= 184.2 g/mol) is one of the simplest of these drugs. What is the boiling point of a solution prepared by dissolving 42.5 g of barbital in 825 g of acetic acid? Kb = 3.07°C/m; boiling point of pure acetic acid = 117.9°C
A) 117.0°C
B) 117.7°C
C) 118.1°C
D) 118.8°C
E) >120°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 7.112 M solution of sulfuric acid (H2SO4) in water has a density of 1.395 g/mL. What is its molality?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mole fraction of potassium nitrate in an aqueous solution is 0.0194. The solution's density is 1.0627 g/mL. Calculate the molarity of the solution.
A) 0.0194 M
B) 0.981 M
C) 1.05 M
D) 1.96 M
E) 19.4 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What volume of concentrated (14.7 M) phosphoric acid is needed to prepare 25.0 L of 3.0 M H3PO4?
A) 0.20 L
B) 0.57 L
C) 1.8 L
D) 3.6 L
E) 5.1 L
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the freezing point of a solution made by dissolving 3.50 g of potassium chloride ( 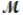 = 74.55 g/mol) in 100.0 g of water. Assume ideal behavior for the solution; Kf = 1.86°C/m.
= 74.55 g/mol) in 100.0 g of water. Assume ideal behavior for the solution; Kf = 1.86°C/m.
A) -1.7°C
B) -0.9°C
C) 0.0°C
D) 0.9°C
E) 1.7°C
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 105
Related Exams