A) acetic acid
B) butyric acid
C) caproic acid
D) formic acid
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be produced as the result of a dehydration reaction between a carboxylic acid and an alcohol?
A) aldehyde
B) ester
C) acid anhydride
D) alkene
F) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Pure liquid carboxylic acids are strongly hydrogen bonded even in the absence of water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the product of the following reaction? 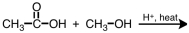
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Two molecules of a carboxylic acid are joined together by two hydrogen bonds.This is referred to as a(n) ____.
A) dimer
B) amide
C) ester
D) carboxylate
F) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Butyric acid is composed of a molecule that is small enough to evaporate from the liquid state at room temperature and,therefore,may have a scent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following has the greatest solubility in water?
A) formic acid
B) propionic acid
C) acetic acid
D) all are equal
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets of reactants could be used to produce a polyester?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the common name of the compound shown below? 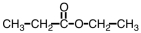
A) methyl propionate
B) propyl acetate
C) ethyl acetate
D) ethyl propionate
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As CH3COOH dissolves in water,H3O+ and ____ are formed.
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would be the products if ethyl stearate were to undergo hydrolysis?
A) There would be no products formed.
B) The products would be ethylaldehyde and stearone.
C) The products would be ethanol and stearic acid.
D) The products would be ethyl stearate hydrate.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about carboxylic acids is true?
A) All are weak acids.
B) Produce hydrogen ions in water.
C) Will react with strong bases.
D) All of the choices are true.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Esters are more easily prepared from acid chlorides than from the acid directly.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which term correctly describes the following reaction? 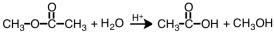
A) esterification
B) dissociation
C) hydrolysis
D) saponification
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many mL of a 0.150 M solution of KOH would be required to titrate 25.0 mL of a 0.125 M solution of the following acid,which can be used as a standard in acid-base titrations? 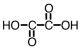
A) 10.4 mL
B) 30.0 mL
C) 20.8 mL
D) 41.7 mL
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following conditions would favor the carboxylate ion instead of the carboxylic acid?
A) low pH
B) high pH
C) both low pH and high pH
D) neither low pH or high pH
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is structure of bromomethyl ethanoate?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following ion could be converted to the corresponding carboxylic acid by _____. 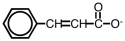
A) heating
B) adding an acid
C) adding water
D) adding a base
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If an intermediate in metabolism is a diester of phosphoric acid,it would have which general formula? (Note: R represents a generic alkyl chain. )
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Saponification is a chemical reaction
A) that results in a soap.
B) due to the presence of a strong organic acid.
C) that results in an alcohol and a carboxylic acid.
D) that explains why soap does not wash off easily in hard water.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 86
Related Exams