Correct Answer

verified
Correct Answer
verified
Short Answer
What kind of molecular orbital (σ, σ*, π, or π*)results when the two atomic orbitals shown below interact in the manner indicated? 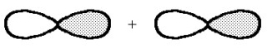
Correct Answer

verified
Correct Answer
verified
Essay
Draw the shape of a 2p orbital.
Correct Answer

verified
Correct Answer
verified
Short Answer
Ar, K+, Cl- are isoelectronic elements (elements with the same number of electrons). What orbital does the last electron occupy?
Correct Answer

verified
3p orbital
Correct Answer
verified
Multiple Choice
Give the H-C-H bond angle in H2CO.
A) 60
B) 90
C) 109.5
D) 120
E) 180
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules does not exhibit a net dipole moment of zero?
A) CO2
B) CH4
C) CCl4
D) H2O
E) SO3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What are the formal charges on nitrogen and the starred oxygen atom in the following molecule? 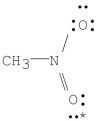
A) N = -1, O = 0
B) N = +1, O = -1
C) N = +1, O = +1
D) N = -1, O = -1
E) N= +1, O = 0
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The N-H single bond in methyl amine (CH3NH2) is a ________ bond formed by the overlap of a(n) ________ orbital on N and a(n) ________ orbital on H.
A) σ; sp2; s
B) σ ; sp3; s
C) π ; sp3; s
D) π ; sp2; p
E) π ; p ; p
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements correctly describes the third electron shell that surrounds the nucleus of an atom?
A) The third shell contains only s and p atomic orbitals.
B) The maximum number of electrons that can occupy the third shell is 18.
C) The total number of atomic orbitals present in the third shell is 16.
D) The third shell can contain f orbitals.
E) All third shell elements must have d electrons.
G) C) and E)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Identify the compound(s) that have a nonzero dipole moment.
A) CH3NH2
B) CO2
C) CH3OCH3
D) (CH3) 2C ![]() C(CH3) 2
C(CH3) 2
E) BCl3
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the CNN bond angle in the compound shown below? 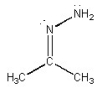
A) ~60°
B) ~90°
C) ~110°
D) ~120°
E) ~180°
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The hydrogen-halogen bond becomes ________ and ________ as the size (atomic weight) of the halogen increases.
A) longer, weaker
B) longer, stronger
C) shorter, weaker
D) shorter, stronger
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the electronic configuration for N-3.
A) 1s22s2
B) 1s22s22p3
C) 1s22s22p4
D) 1s22s22p6
E) 1s22s22p63s1
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Draw the Lewis structure for CH3N2+.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Triethylamine [(CH3CH2) 3N] is a molecule in which the nitrogen atom is ________ hybridized and the CNC bond angle is ________.
A) sp2, >109.5°
B) sp2, <109.5°
C) sp3, >109.5°
D) sp3, <109.5°
E) sp, 109.5°
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many sp2 hybridized carbons are present in allene (H2C=C=CH2) ?
A) 0
B) 1
C) 1) 5
D) 2
E) 3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Using the symbol δ+and δ-, show the direction of the polarity in the indicated bond. 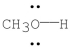
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the interaction of two hydrogen 1s atomic orbitals of the same phase. Which of the statements below is an incorrect description of this interaction?
A) A sigma bonding molecular orbital is formed.
B) The molecular orbital formed is lower in energy than a hydrogen 1s atomic orbital.
C) The molecular orbital formed has a node between the atoms.
D) The molecular orbital formed is cylindrically symmetric.
E) A maximum of two electrons may occupy the molecular orbital formed.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Essay
Draw the Kekulé structure and show the direction of the dipole moment for CH2Cl2.
Correct Answer

verified
11ea7d6e_065f_12e3_8568_a34d445fb275_TB1830_00_TB1830_00
Correct Answer
verified
Essay
Draw condensed structures for the four compounds with formula C3H9N.
Correct Answer

verified
CH3CH2CH2NH2
C...View Answer
Show Answer
Correct Answer
verified
C...
View Answer
Showing 1 - 20 of 81
Related Exams