B) False
Correct Answer

verified
Correct Answer
verified
True/False
The rates of radioactive decay processes, like the rates of chemical reactions, are sensitive to temperature changes.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What element is the stable end-product of the uranium radioactive decay series?
A) Th
B) Pu
C) Ra
D) Au
E) Pb
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A rock contains 0.37 mg of Pb-206 and 0.95 mg of U-238. The half-life of the decay series U-238 Pb-206 is 4.5 109 yr. Assuming no Pb-206 was present in the rock initially, how old is the rock?
A) 1.7 109 yr
B) 5.2 109 yr
C) 2.7 106 yr
D) 4.5 109 yr
E) 2.4 109 yr
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine how much energy is released when polonium-210 decays according to ![Determine how much energy is released when polonium-210 decays according to . [Atomic masses: polonium-210 = 209.982857 amu; helium-4 = 4.002603 amu; lead-206 = 205.974449 amu] A) 4.14 \times 10<sup>9</sup> kJ/mol B) 7.20 \times 10<sup>11</sup> kJ/mol C) 5.22 \times 10<sup>8</sup> kJ/mol D) 4.66 \times 10<sup>9</sup> kJ/mol E) 6.43 \times 10<sup>12</sup> kJ/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3245/11ea7cc5_01d1_8045_a2ab_f5090605275f_TB3245_11.jpg) . [Atomic masses: polonium-210 = 209.982857 amu; helium-4 = 4.002603 amu; lead-206 = 205.974449 amu]
. [Atomic masses: polonium-210 = 209.982857 amu; helium-4 = 4.002603 amu; lead-206 = 205.974449 amu]
A) 4.14 109 kJ/mol
B) 7.20 1011 kJ/mol
C) 5.22 108 kJ/mol
D) 4.66 109 kJ/mol
E) 6.43 1012 kJ/mol
G) C) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The mass of a nucleus is always less than the sum of the masses of the nucleons.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Gamma-rays cause radiation damage when they interact with matter by producing
A) ions and free radicals.
B) isotopes.
C) daughter products.
D) oxidation.
E) reduction.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
The isotope of iodine with mass number 128 undergoes beta decay with a half-life of 25.00 minutes. How long will it take for 93.75% of a sample of pure 128 I to decay?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What would the atom ratio of 206Pb to 238U be in a uranium mineral from a rock that is 1.0 109 years old? t1/2(238U) = 4.5 109 yr.
A) 0.14
B) 0.16
C) 0.22
D) 0.86
E) 1.16
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Balance the equation 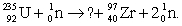
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine how much energy is released when thorium-230 decays according to ![Determine how much energy is released when thorium-230 decays according to . [Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu] A) 3.98 \times 10<sup>9</sup> kJ/mol B) 4.60 \times 10<sup>8</sup> kJ/mol C) 7.20 \times 10<sup>11</sup> kJ/mol D) 4.90 \times 10<sup>9</sup> kJ/mol E) 7.15 \times 10<sup>11</sup> kJ/mol](https://d2lvgg3v3hfg70.cloudfront.net/TB3245/11ea7cc5_01d1_8044_a2ab_f5e5d54af373_TB3245_11.jpg) . [Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu]
. [Atomic masses: thorium-230 = 230.033127 amu; helium-4 = 4.002603 amu; radium-226 = 226.025403 amu]
A) 3.98 109 kJ/mol
B) 4.60 108 kJ/mol
C) 7.20 1011 kJ/mol
D) 4.90 109 kJ/mol
E) 7.15 1011 kJ/mol
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an advantage of nuclear power plants over coal-burning plants? Nuclear power plants
A) produce radioactive byproducts with very short half-lives, reducing the need for waste storage.
B) do not pollute the air with SO2, soot, and fly-ash.
C) create no thermal pollution.
D) generate radioactive byproducts that can be sold for use in secondary applications.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The only stable isotope of fluorine is 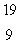 F. What type of radioactivity would you expect from the isotope
F. What type of radioactivity would you expect from the isotope 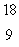 F, which has one fewer neutron?
F, which has one fewer neutron?
A) ![]() p
p
B) ![]() n
n
C) ![]()
D) ![]()
E) ![]() He
He
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Complete and balance the nuclear equation 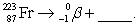
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Rb-87/Sr-87 method of dating rocks is often used by geologists: 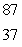 Rb
Rb 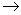
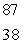 Sr +
Sr + 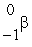 t1/2 = 6.0 1010 yr Estimate the age of a rock sample in which the present-day mole ratio of Rb-87 to Sr-87 is 36:1.
t1/2 = 6.0 1010 yr Estimate the age of a rock sample in which the present-day mole ratio of Rb-87 to Sr-87 is 36:1.
A) 2.4 109 yr
B) 1.7 109 yr
C) 3.1 1011 yr
D) 4.1 10-11 yr
E) 3.6 1011 yr
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
Strontium-90 has a half-life of 28.8 years.How much strontium-90 was present initially, if after 144 years 10.0 g remain?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The only stable isotope of aluminum is aluminum-27. What type of radioactive decay should be expected from 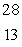 Al?
Al?
A) ![]() H
H
B) ![]() n
n
C) ![]()
D) ![]()
E) ![]() He
He
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The heaviest known isotope of hydrogen is called tritium, 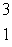 H. It decays by beta emission, and has a half-life of 12.3 years. What fraction of a tritium sample will remain after 5.20 years?
H. It decays by beta emission, and has a half-life of 12.3 years. What fraction of a tritium sample will remain after 5.20 years?
A) 0.0210
B) 0.746
C) 3.41
D) 0.254
E) 0.423
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which isotope, when bombarded with nitrogen-15, yields the artificial isotope dubnium-260 plus 4 neutrons?
A) californium-245
B) thorium-257
C) nobelium-245
D) californium-249
E) dubnium-249
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
A plot of the number of neutrons versus the number of protons in various isotopes produces a "belt of stability." Isotopes below the belt of stability (i.e., with a smaller neutron-to-proton ratio)decay by beta particle emission.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 112
Related Exams