A) two monodentate ligands and two bidentate ligands.
B) three monodentate ligands and two bidentate ligands.
C) four monodentate ligands and two bidentate ligands.
D) one monodentate ligand and four bidentate ligands.
E) four bidentate ligands.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound [Pt(NH3) 2Cl2], the coordination number and oxidation number of the central atom are, respectively,
A) 2, 0.
B) 4, 4.
C) 5, 0.
D) 4, 2.
E) 6, 2.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the oxidation number of Fe in [Fe(CN)6]4-?
Correct Answer

verified
Correct Answer
verified
Essay
Give (a)the oxidation number of the metal, (b)the number of d electrons, (c)the coordination number, (d)the charge of the complex ion, and (e)the number and type of ligands for the coordination compound shown below. 
Correct Answer

verified
(a)+3, (b)3 d electr...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The electron configuration of a Cr3+ ion is
A) [Ar]3d5.
B) [Ar]4s13d2.
C) [Ar]3d3.
D) [Ar]4s13d5.
E) [Ar]4s23d4.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the complex ion [ML6]n+, Mn+ has five d electrons and L is a strong field ligand.According to crystal field theory, the magnetic properties of the complex ion correspond to how many unpaired electrons?
A) 0
B) 1
C) 2
D) 3
E) 5
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of Co3+ is
A) 4
B) 5
C) 6
D) 7
E) 10
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The total number of electrons in the 3d orbitals of a copper atom is
A) 6
B) 7
C) 8
D) 9
E) 10
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The best name for the complex shown below is 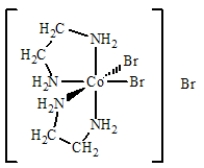
A) cobalt(III) bis(ethylenediamine) bromide.
B) dibromobis(ethylenediamine) cobalt(III) bromide.
C) dibromidedi(ethylenediamine) cobalt(III) bromide.
D) dibromodiethylenediaaminecobalt(III) bromide.
E) tribromobis(ethylenediamine) cobalt(III) .
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these square planar complex ions can have cis-trans isomers?
A) [Pt(NH3) 4]2+
B) [Ni(NH3) 4]2+
C) [Pt(NH3) 2Cl2]
D) [Pt(NH3) Cl3]-
E) [Ni(NH3) 3Cl]+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cis-trans isomerism is exhibited by which one of these ions?
A) [Pd(NH3) 3Cl]+
B) [Co(NH3) 5Cl]2+
C) [Fe(CN) 6]3-
D) All of the above.
E) None of the above.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the coordination number of cobalt in [Co(NH3)6]Cl3?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which response gives the correct coordination number (C.N.) and oxidation number (O.N.) of the transition metal atom in [Co(NH3) 2(H2O) 2Cl2]+?
A) C.N.= 2; O.N.= +3
B) C.N.= 3; O.N.= +1
C) C.N.= 4; O.N.= +2
D) C.N.= 6; O.N.= +1
E) C.N.= 6; O.N.= +3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the coordination compound K2[Co(en) Cl4], the coordination number (C.N.) and oxidation number (O.N.) of cobalt are
A) C.N.= 6; O.N.= +2.
B) C.N.= 6; O.N.= +3.
C) C.N.= 5; O.N.= +2.
D) C.N.= 5; O.N.= +4.
E) C.N.= 4; O.N.= +3.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these ligands produces the weakest crystal field?
A) CN-
B) I-
C) OH-
D) H2O
E) NH3
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The electron configuration of a Ti atom is
A) [Ne]3s23d2.
B) [Ne] 3s24d2.
C) [Ar]4s23d2.
D) [Ar]4s24d2.
E) [Ar]3d4.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Essay
Write the chemical formula of triammineaquodichlorochromium(III)chloride.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the complex ion [Cr(C2O4) 2(H2O) 2]-, the oxidation number of Cr is
A) +1.
B) +2.
C) +3.
D) -2.
E) -1.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Cis-platinum complexes are
A) used in the extraction of silver and gold.
B) effective antidotes for heavy metal poisoning (e.g., Pb2+ and Hg2+) .
C) used to provide nutrients for plants.
D) used to prevent eutrophication of lakes.
E) effective antitumor agents.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in the complex ion [Mn(CN) 6]3-?
A) 0
B) 1
C) 2
D) 3
E) 4
G) All of the above
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 80
Related Exams