A) 42.7 kJ
B) 49.5 kJ
C) 179 kJ
D) 1970kJ
E) 6840 kJ
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms does not participate in hydrogen bonding?
A) S
B) O
C) F
D) N
F) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Suppose the atoms in a two-dimensional crystal have the following arrangement: 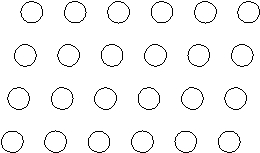 How many atoms are in one unit cell?
How many atoms are in one unit cell?
Correct Answer

verified
one (each corner of ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
The triple point of iodine is at 0.12 atm and 115°C. Thus, liquid I2
A) is more dense than I2 (s) .
B) cannot exist above 115°C.
C) is liquid at room temperature.
D) cannot have a vapor pressure less than 91 torr.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The freezing point of a liquid does not change as the atmospheric pressure changes.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following substances would have the highest critical temperature?
A) CH3Cl
B) C2H6
C) F2
D) H2
E) CO2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The molar heats of sublimation and fusion of iodine are 62.3 kJ/mol and15.3 kJ/mol, respectively. Calculate the molar heat of vaporization of liquid iodine.
A) 77.6 kJ/mol
B) 47.0 kJ/mol
C) -47.0 kJ/mol
D) -77.6 kJ/mol
E) 4.07 kJ/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The shape of the water-to-glass meniscus results from the strong adhesive forces between glass and water.
B) False
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest)type of intermolecular force present in RbCl(s).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following properties indicates the presence of weak intermolecular forces in a liquid?
A) a high heat of vaporization
B) a high critical temperature
C) a high vapor pressure
D) a high boiling point
E) None of the above.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Indicate all the types of intermolecular forces of attraction in CF4(g).
Correct Answer

verified
Correct Answer
verified
Short Answer
Of the given pair of compounds, which would have the higher boiling point? NH3 or CH4
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following elements would have the lowest melting point?
A) Kr
B) Br2
C) S8
D) Ca
E) K
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be expected to have the highest vapor pressure at room temperature?
A) ethanol, bp = 78°C
B) methanol, bp = 65°C
C) water, bp = 100°C
D) acetone, bp = 56°C
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following substances should exhibit hydrogen bonding in the liquid state?
A) SiH4
B) H2
C) H2S
D) CH4
E) CH3NH2
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Methane has a heat of fusion of 0.84 kJ/mol and a heat of vaporization of 9.2 kJ/mol.Estimate the value for the heat of sublimation.
Correct Answer

verified
Correct Answer
verified
Essay
The meniscus for water is curved upward at the edges (i.e., it is "concave up"). Explain this phenomenon in terms of cohesion and adhesion.
Correct Answer

verified
Adhesion of water to...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Crystals of elemental sulfur are easily crushed, and melt at 113°C.Liquid sulfur does not conduct electricity.What kind of crystal is this?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Vanadium crystallizes in a body-centered cubic lattice, and the length of the edge of a unit cell is 305 pm. What is the density of V?
A) 5.96 g/cm3
B) 2.98 g/cm3
C) 2.98 * 10-6 g/cm3
D) 5.96 * 10-30 g/cm3
E) 11.9 g/cm3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following compound and its boiling point, identify whether it is polar or nonpolar: Ar, -185.7°C.
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 140
Related Exams