Correct Answer

verified
Correct Answer
verified
Short Answer
What bond angles are predicted by VSEPR theory for the F -P -F bonds in PF5?
Correct Answer

verified
Correct Answer
verified
Short Answer
Ozone (O3)is an allotropic form of oxygen. Use VSEPR theory to predict the shape of the ozone molecule.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the iodine atom in the IF5 molecule?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The F - S - F bond angles in SF6 are
A) 90° and 180°
B) 109.5°
C) 120°
D) 180°
E) 90° and 120°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, the geometry of the PH3 molecule is best described as
A) linear.
B) trigonal planar.
C) tetrahedral.
D) bent.
E) trigonal pyramidal.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules have the same geometries?
A) SF4 and CH4
B) CO2 and H2O
C) CO2 and BeH2
D) N2O and NO2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The F -Cl -F bond angles in ClF3 are expected to be approximately
A) 90° only.
B) 109.5° only.
C) 120° only.
D) 180° only.
E) 90° and 180°.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Explain why CO2 is nonpolar, but OCS is polar.
Correct Answer

verified
In CO2 the two bond moments point in oppo...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
N,N-diethyl-m-tolumide (DEET) is the active ingredient in many mosquito repellents.What is the hybridization state of carbon indicated by the arrow in the structure of DEET shown below? 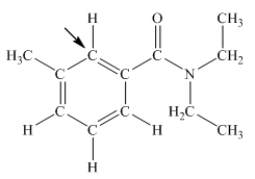
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which one of the following molecules is the central atom sp2 hybridized?
A) SO2
B) N2O
C) BeCl2
D) NF3
E) PF5
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the species Cl2+, Cl2, and Cl2-. Which of these species will be paramagnetic?
A) only Cl2
B) Cl2+ and Cl2
C) Cl2 and Cl2-
D) Cl2+ and Cl2-
E) all three are paramagnetic
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the molecular geometry of SeF4.
A) 0 lone pairs, tetrahedral
B) 1 lone pair, distorted tetrahedron (seesaw)
C) 1 lone pair, square pyramidal
D) 1 lone pair, tetrahedral
E) 2 lone pairs, square planar
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the geometry around the central atom in XeO4.
A) trigonal planar
B) trigonal pyramidal
C) tetrahedral
D) trigonal bipyramidal
E) octahedral
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The number of pi bonds in the molecule below is 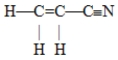
A) 1
B) 2
C) 3
D) 5
E) 9
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in CCl4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR theory, which one of the following species should be linear?
A) H2S
B) HCN
C) BF3
D) H2CO
E) SO2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the geometry of the ion IBr2-.
A) 0 lone pairs, linear
B) 1 lone pair, bent
C) 2 lone pairs, bent
D) 3 lone pairs, bent
E) 3 lone pairs, linear
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The geometry of the ClF3 molecule is best described as:
A) distorted tetrahedron
B) trigonal planar
C) tetrahedral
D) T-shaped
E) trigonal pyramidal
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of lone pairs around the central atom and the molecular geometry of SCl2.
A) 0 lone pairs, linear
B) 1 lone pair, bent
C) 2 lone pairs, bent
D) 3 lone pairs, bent
E) 3 lone pairs, linear
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 122
Related Exams