Correct Answer

verified
11ea7cbf_8f08_aef8_a2ab_73332304535d_TB3246_00
Correct Answer
verified
Multiple Choice
The Lewis dot symbol for the S 2- ion is
A) ![]()
B) ![]() 2-
2-
C) S2-
D) - ![]() - 2-
- 2-
E) ![]() -
-
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound?
A) ClF3
B) FeCl3
C) NH3
D) PF3
E) SO3
G) B) and D)
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
The number of resonance structures for the nitrate ion that satisfy the octet rule is
A) 1
B) 2
C) 3
D) 4
E) None of these
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Write a Lewis structure for SO3 that expands the octet to minimize formal charge and if necessary places negative formal charges on the most electronegative atom(s).
Correct Answer

verified
Correct Answer
verified
True/False
Of the species NO2, NO, and N2, only NO2 is an exception to the octet rule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Assuming the octet rule is obeyed, how many covalent bonds will an oxygen atom form to give a formal charge of zero?
A) 0
B) 1
C) 2
D) 3
E) 4
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete this statement: Coulomb's law states that the magnitude of the force of interaction between two charged bodies is
A) directly proportional to the product of the charges on the bodies and directly proportional to the distance separating them.
B) directly proportional to the product of the charges on the bodies, and inversely proportional to the square of the distance separating them.
C) inversely proportional to the product of the charges on the bodies, and directly proportional to the square of the distance separating them.
D) directly proportional to the sum of the charges on the bodies, and inversely proportional to the square of the distance separating them.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the Born-Haber cycle to calculate the lattice energy of MgO (s) given the following data: H(sublimation) Mg = 130 kJ/mol
I1 (Mg) = 738.1 kJ/mol
I2 (Mg) = 1450 kJ/mol
Bond energy (O=O) = 498.7 kJ/mol
EA (O) = 141 kJ/mol
EA (O-) = -780 kJ/mol
H 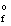 (MgO(s) ) = -601.8 kJ/mol
(MgO(s) ) = -601.8 kJ/mol
A) 2200 kJ/mol
B) 2800 kJ/mol
C) 3200 kJ/mol
D) 3800 kJ/mol
E) 4100 kJ/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The azide ion, N3-, is very reactive although it is isoelectronic with the very stable CO2 molecule.This reactivity is reasonable considering that
A) a Lewis structure cannot be written for the azide ion that has nitrogen formal charges of zero.
B) there is no valid Lewis structure possible for the azide ion.
C) there are resonance structures for azide ion but not for carbon dioxide.
D) nitrogen cannot form multiple bonds.
E) charged species always decompose in solution.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solids would have the highest melting point?
A) NaI
B) NaF
C) MgO
D) MgCl2
E) KF
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
A molecule with two resonance structures is shifting quickly back and forth from one structure to the other
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is most likely to be an ionic compound?
A) NCl3
B) BaCl2
C) CO
D) SO2
E) SF4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in order of increasing ionic character 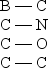
A) B - C < C - N < C - O < C - C
B) C - C < B - C < C - N < C - O
C) C - C < C - N < B - C < C - O
D) C - O < C - N < B - C < C - C
E) C - N < C - C < B - C < C - O
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What type of bonding is present in the molecule NH2NH2 (ionic, covalent, or both)?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the elements C, O, and H in order of increasing electronegativity
A) C < O < H
B) H < C < O
C) C < H < O
D) O < C < H E H < O < C
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
List all types of bonding present in the molecule SO3 I.ionic bond II.polar covalent bond III.nonpolar covalent bond
A) I only
B) II only
C) III only
D) I and II
E) II and III
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in order of increasing ionic character 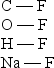
A) C - F < O - F < H - F < Na - F
B) C - F < H - F < O - F < Na - F
C) O - F < C - F < H - F < Na - F
D) H - F < C - F < O - F < Na - F
E) Na - F < H - F < C - F < O - F
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
BeF42- is called the fluoberyllate ion.The formal charge on the beryllium atom in this ion is
A) -2
B) -1
C) 0
D) +1
E) +2
G) A) and B)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Use bond energies to estimate the enthalpy of formation of HBr(g) . BE(H-H) = 436 kJ/mol BE(Br-Br) = 192 kJ/mol BE(H-Br) = 366 kJ/mol
A) +262 kJ/mol
B) +104 kJ/mol
C) +52 kJ/mol
D) -52 kJ/mol
E) -104 kJ/mol
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 137
Related Exams