A) potential,kinetic
B) kinetic,potential
C) kinetic,free
D) potential,stored
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
ATP is considered a high-energy compound because under cellular conditions,7.3 kcal per mole of energy is released when a bond is broken between:
A) the base adenine and the sugar ribose.
B) the adenosine and the phosphate groups.
C) the base adenine and the phosphate groups.
D) the adenosine diphosphate and the third phosphate.
E) All of the bonds release energy as ATP is completely broken down.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
An enzyme is a globular protein that inhibits the formation of chemical bonds within the enzyme's substrate(s)causing the rate of the reaction to slow down.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The energy for ATP synthesis in chemiosmotic phosphorylation comes from the movement of hydrogen ions across a membrane down a concentration gradient.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is consistent with the laws of physics governing energy?
A) When a liter of gasoline is burned in a car engine,100% of its energy goes into moving the car along the road.
B) You eat a "quarter-pounder" hamburger and assemble exactly a quarter-pound of additional body weight on your body.
C) Eventually sunlight that is absorbed on the earth returns to space as dispersed heat.
D) A calorie of sunlight becomes a calorie of plant tissue that,eaten by you,becomes a calorie of heat lost in muscle "power."
E) Chemical bonds are a case of converting energy to matter; breaking the bonds converts matter to energy.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the electron transport systems of chloroplasts and mitochondria,
A) the system consists of a series of membrane bound carriers that transfer electrons from one carrier to another.
B) high energy electrons enter the system and low energy electrons exit the system.
C) energy release occurs when the electron transfers from one carrier to another.
D) All of the choices are correct.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
If a change in pH alters an allosteric site where an inhibitor binds,but doesn't change the active site for the intended substrate,it would be possible for an enzymatically controlled reaction to occur as normal.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In this pathway,B C is coupled with ADP ATP.Categorize the reactions as endergonic or exergonic. 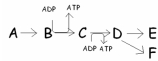
A) B C is endergonic and ADP
ATP is exergonic.
B) ADP ATP is endergonic and B
C is exergonic.
C) Both B C and ADP
ATP are endergonic.
D) Both B C and ADP
ATP are exergonic.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
ATP is considered to be
A) an enzyme used widely in all kinds of cells.
B) a coenzyme used to inhibit or activate different enzymes.
C) a molecule that carries a great deal of chemical energy in a chemical bond.
D) the precursor of a high-energy membrane-bounded protein.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
During the conversion of glucose into a free form of energy only a small percentage is converted into useable ATP.What is the rest of the energy converted into?
A) heat
B) CO2
C) H2O
D) CO-
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Study the figure at right.What does the letter A depict? 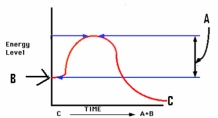
A) Energy of the reactant
B) Energy of the products
C) Energy of activation
D) Substrate concentration
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following labels is incorrectly identified. 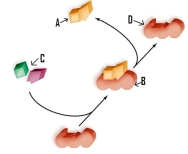
A) A refers to the products.
B) B refers to the substrate-enzyme complex.
C) C refers to the reactants.
D) D refers to the product.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement describes the currently accepted theory of how an enzyme and its substrate fit together?
A) As the product is released,the enzyme breaks down.
B) The enzyme is like a key that fits into the substrate,which is like a lock.
C) The active site is permanently changed by its interaction with the substrate.
D) As the substrate binds to the enzyme,the shape of the active site changes to accommodate the reaction.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which statement is NOT true about how various conditions will effect the activity of an enzyme?
A) Higher temperatures generally increase the activity of an enzyme up to a point.
B) Above a certain range of temperatures,the protein of an enzyme is denatured.
C) A change in pH can cause an enzyme to be inactivated.
D) An enzyme's activity is generally reduced by an increase in substrate concentration.
E) When sufficient substrate is available,the active site will nearly always be occupied.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An automobile engine is about 20 - 30% efficient in converting chemical energy to mechanical energy.Cells are about 39% efficient in the transformation of glucose to ATP.The rest of the energy is lost as heat.This is illustrative of the:
A) First Law of Thermodynamics.
B) Second Law of Thermodynamics.
C) Third Law of Thermodynamics.
D) The Cell Theory.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Endergonic reactions:
A) release energy.
B) have a negative G and occur spontaneously.
C) can only occur if there is an input of energy.
D) have products with less free energy than the reactants.
E) All of the choices are correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these statements is NOT a consequence of the second law of thermodynamics?
A) While the total amount of energy is unchanged,the energy lost as heat is no longer useful to the cell in doing work.
B) Reactions that occur spontaneously are those that increase the amount of useful energy in a system.
C) The amount of disorder in the universe is always increasing.
D) To maintain organization of a cell,a continual input of energy is required.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Astrophysicists explain that eventually the sun will swell to become a red giant,engulf the earth and "burn out" with all forms of energy dispersing in a final "heat death." Compared with conditions today,the entropy of the universe then will
A) have increased greatly.
B) have decreased greatly.
C) remain the same because energy cannot be created or destroyed.
E) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Study the graph at right.The letter 'B ' depicts the _____,while 'C' depicts ________. 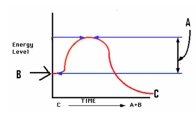
A) Energy of Activation; Energy of products
B) Energy of products; Energy of Activation
C) Energy of reactants; Energy of products
D) Reactant concentration; Activation Energy
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Coupling occurs when the energy released by an exergonic reaction is:
A) used to drive another exergonic reaction.
B) used to drive an endergonic reaction.
C) lost as nonusable heat to the environment.
D) used to decrease the entropy of the universe.
E) All of the choices are correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 55
Related Exams