A) 4N3-, 12H+, PO42-
B) 3NH4+, 4PO43-
C) N3-, 12H+, P3- ; 4O2-
D) 3NH4+, PO43-
E) 4NH+, 3PO43-
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the examples in which the formulas do not correctly match the names of the compounds indicated. 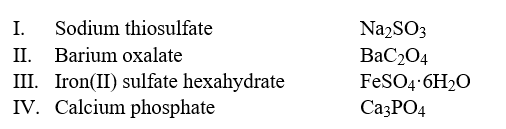 Hint: Write the correct name of each formula without looking at the names and then compare it to the choices.
Hint: Write the correct name of each formula without looking at the names and then compare it to the choices.
A) II only
B) II and III
C) I, II and IV
D) I and IV
E) II, III and IV
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the formula of the compound formed from the calcium ion and HCO3-?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Diamond and graphite are different forms of which element?
A) sodium
B) carbon
C) mercury
D) gold
E) calcium
G) All of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the formula of the alkane hydrocarbon with seven carbon atoms.
Correct Answer

verified
Correct Answer
verified
Short Answer
The symbol Te belongs to a metalloid in group ________.
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the formula for the compound that has the atoms and, or groups in the order given: 3 Fe, and two groups made up of 1 As and 4 O. Hint: The groups of atoms are polyatomic ions.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name of the compound, HCN(g) ?
A) hydrocarbonitride
B) hydrocyanic acid
C) hydrogen carbonitride
D) hydrogen cyanate
E) hydrogen cyanide
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the best name for the I2O5 molecule?
A) diiodine pentoxide
B) iodine pentoxygen
C) pentoxygen iodide
D) iodine dioxide
E) diiodide oxide
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The formula for magnesium phosphide is Mg3P2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many atoms of each element appear on each side of the arrow in the following chemical equation? 3Cl3BNH2CH3 + 6(CH3) 3N 6(CH3) 3NHCl + B3N3Cl3(CH3) 3 Hint: Distribute coefficients across the entire compound for each reactant and product. Be sure to account for parenthesis.
A) 9Cl, 3B, 9N, 54H, 6C
B) 9Cl, 3B, 9N, 69H, 21C
C) 3Cl, 3B, 9N, 15H, 21C
D) 9Cl, 1B, 9N, 54H, 9C
E) 3Cl, 3B, 9N, 69H, 18C
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The name of the compound Al(SO4) 3 is
A) aluminum sulfide.
B) aluminum sulfate.
C) aluminum trisulfate.
D) aluminum(III) sulfate.
E) aluminum sulfite.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements has the most similar chemical properties to sulfur?
A) calcium
B) oxygen
C) phosphorus
D) bromine
E) nitrogen
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The alkali metals like sodium and potassium are soft metals, so they are unreactive towards water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements has the most similar chemical properties silicon?
A) aluminum
B) phosphorus
C) nitrogen
D) silver
E) germanium
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound is correctly represented as a hydrate?
A) C2H5OH2+
B) MgSO4·7H2O
C) FeH2(CO) 4
D) O2S(OH) 2
E) [CrCl(H2O) 5]Cl
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Which group of nonmetallic elements is called "inert"?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of these elements has the most similar chemical properties to magnesium?
A) calcium
B) sodium
C) aluminum
D) iron
E) cesium
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound exists as a diatomic molecule in its free state?
A) magnesium
B) manganese
C) silicon
D) arsenic
E) chlorine
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the name for the compound V2O5?
A) divanadium pentoxide
B) vanadic oxide
C) vanadium(V) oxide
D) vanadium(V) pentoxide
E) vanadous oxide
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 227
Related Exams