B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An ideal gas (the system) is contained in a flexible balloon at a pressure of 1 atm and is initially at a temperature of 20.°C. The surrounding air is at the same pressure, but its temperature is 25°C. When the system has equilibrated with its surroundings, both system and surroundings are at 25°C and 1 atm. In changing from the initial to the final state, which one of the following relationships regarding the system is correct?
A) ΔU < 0
B) ΔU = 0
C) ΔH = 0
D) w > 0
E) q > 0
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system which does work on the surroundings with no heat change has
A) w = ΔU.
B) w = -ΔU.
C) w > 0, ΔU < 0.
D) w < 0, ΔU > 0.
E) w > ΔU.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using Hess's law, what is ΔH°rxn for the following reaction? WO3(s) + 3H2(g) → W(s) + 3H2O(g) 
A) 117.5 kJ/mol
B) 235.0 kJ/mol
C) 359.3 kJ/mol
D) 1202.2 kJ/mol
E) 2169.4 kJ/mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the equation for the formation reaction for gaseous hydrazine, N2H4, at 25ºC and 1 atm pressure?
A) 2N2H4(g) → 2NH3(g) + H2(g)
B) 2NH3(g) + H2(g) → N2H4(g)
C) N2(g) + 2H2O(g) → N2H4 (g) + O2(g)
D) N2(g) +2H2(g) → N2H4(g)
E) 2NO2(g) + 6H2(g) → N2H4(g) + 4H2O(g)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much heat is required to raise the temperature of 1500 g of water from 25°C to 52°C? The specific heat of water is 4.184 J/g·°C.
A) 1500 kJ
B) 170 kJ
C) 6.3 kJ
D) 41 J
E) 41 kJ
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Using Hess' law, what is ΔH°rxn at 25°C for the following reaction? ClF(g) + F2(g) → ClF3(g) 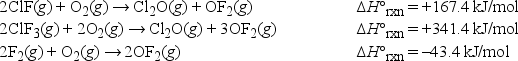
A) -217.5 kJ/mol
B) -130.2 kJ/mol
C) 217.5 kJ/mol
D) -108.7 kJ/mol
E) 465.4 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Solid sodium peroxide (Na2O2) reacts with liquid water yielding aqueous sodium hydroxide and oxygen gas. How much heat is released if 327.2 g of oxygen gas is produced from the reaction of sodium peroxide and water under standard-state conditions? 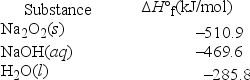
A) 46,630 kJ
B) 1457 kJ
C) 6689 kJ
D) 3345 kJ
E) 2914 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Pentaborane B5H9(s) burns vigorously in O2 to give B2O3(s) and H2O(l) . What is ΔH° for the combustion of 1 mol of B5H9(s) ? Substance ΔH°f (kJ/mol) B2O3(s) -1273.5 B5H9(s) +73.2 H2O(l) -285.8
A) -1486.1 kJ
B) -1632.5 kJ
C) -4396.7 kJ
D) -4652.85 kJ
E) -9086.1 kJ
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? SiO2(s) + 4HCl(g) → SiCl4(g) + 2H2O(g) 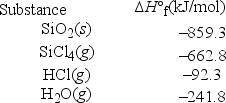
A) -1856.2 kJ/mol
B) -1372.6 kJ/mol
C) -47.0 kJ/mol
D) 82.1 kJ/mol
E) 530.6 kJ/mol
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the following reaction? H3AsO4(aq) + 4H2(g) → AsH3(g) + 4H2O(l) 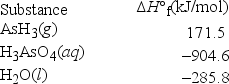
A) -1876.3 kJ/mol
B) -1018.9 kJ/mol
C) -790.3 kJ/mol
D) -410.1 kJ/mol
E) -67.1 kJ/mol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The heat absorbed by a system at constant pressure is equal to ΔU + PΔV.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which equation has a ΔHrxn that is not equal to ΔHof of the product?
A) O2(g) + H2(g) → H2O2(g)
B) ½O2(g) + H2(g) → H2O(g)
C) H2(g) + ½O2(g) + → H2O(l)
D) NO(g) + ½O2(g) → NO2(l)
E) ½H2(g) + ½Cl2(g) → HCl(g)
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Given the following ΔH° values H2(g) + ½O2(g) → H2O(l) ΔH°f = -285.8 kJ H2O2(l) → H2(g) + O2(g) ΔH°rxn = 187.6 kJ calculate ΔH°rxn for the reaction H2O2(l) → H2O(l) + ½O2(g),
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is ΔH°rxn for the reaction 2Al2O3(s) → 4Al(s) + 3O2(g) ? ΔH°f(Al2O3(s) ) = -1670 kJ/mol
A) 3340 kJ/mol
B) 1670 kJ/mol
C) -3340 kJ/mol
D) -1670 kJ/mol
E) -835 kJ/mol
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which reaction would you expect ΔH to be substantially greater than ΔU (i.e., ΔH > ΔU) ?
A) H2(g) + Br2(g) → 2HBr(g)
B) CO2(s) → CO2(g)
C) C2H2(g) + H2(g) → C2H4(g)
D) H2O(s) → H2O(l)
E) HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
A 100.-g sample of Cu is heated to 400.oC and then plunged into 1000. g of water at 22.6oC. What is the final temperature? Specific heat capacity: Cu: 0.385 J/g·°C;water: 4.184 J/g·°C
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction: 2A + B2 → 2AB ΔH = +50.0 kJ.
A) The reaction is endothermic.
B) Heat is released to the surroundings.
C) The standard enthalpy of formation for AB is 50.0 kJ.
D) The bond energy for each A-B bond is 50.0 kJ.
E) The molecule AB contains less energy than A or B2.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system receives 575 J of heat and delivers 424 J of work. What is the change in the internal energy, ΔU, of the system?
A) -150. J
B) 150. J
C) -1000. J
D) 1000. J
E) 575 J
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How much heat is evolved if 0.600 kg of SO2 is burned in excess oxygen? 2SO2(g) + O2(g) → 2SO3(g) ΔH°rxn= -198 kJ/mol
A) 5.46 × 10-2 kJ
B) 927 kJ
C) 1.85 × 103 kJ
D) 59,400 kJ
E) 3.71 × 103 kJ
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 141
Related Exams