A) trigonal planar
B) trigonal pyramidal
C) tetrahedral
D) trigonal bipyramidal
E) octahedral
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to the VSEPR model, the predicted molecular geometry of the SO3 molecule is
A) pyramidal.
B) tetrahedral.
C) trigonal planar.
D) seesaw.
E) square planar.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which has a dipole moment of zero?
A) N2O
B) NF3
C) H2S
D) SeO3
E) CH3Cl
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The bond angle for an sp hybrid orbital is smaller than the bond angle for an sp2 hybrid orbital.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Valence bond theory predicts that iodine will use ________ hybrid orbitals in ICl2-.
A) sp2
B) sp3
C) sp3d
D) sp3d2
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the As atom in the AsF5 molecule?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in CCl4.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which formula is incorrectly matched with its VSEPR model representation? (Note: Lone pairs on the models, if any, are not shown.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of BeH2 as predicted by the VSEPR model?
A) tetrahedral
B) bent
C) trigonal planar
D) linear
E) trigonal pyramidal
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is required for the determination of the VSEPR model and the molecular shape?
A) Atomic mass
B) Number of protons
C) Oxidation number
D) Lewis structure
E) None of the answers is correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following molecules in order of increasing dipole moment. 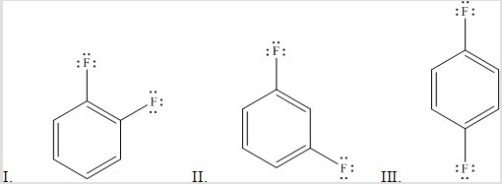
A) III < II < I
B) III < I < II
C) I < II < III
D) I < III < II
E) II < III < I
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When PCl5 solidifies it forms PCl4+cations and PCl6- anions. According to valence bond theory, what hybrid orbitals are used by phosphorus in the PCl4+cation?
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which molecule is the central atom sp2 hybridized?
A) SO2
B) N2O
C) BeCl2
D) NF3
E) PF5
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the most reasonable prediction for the Cl-N-Cl bond angle in NCl3?
A) 120°
B) 111°
C) 109.5°
D) 107°
E) 90°
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry of ClF2- as predicted by the VSEPR model?
A) linear
B) bent
C) seesaw
D) T-shaped
E) trigonal bipyramidal
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
How many π bonds are there in CH3COCH3?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the total number of electron domains around the central atom for a molecule having a trigonal pyramidal molecular geometry, such as NH3?
A) 5
B) 2
C) 3
D) 4
E) 6
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Indicate the type of hybrid orbitals used by the central atom in PCl3.
A) sp
B) sp2
C) sp3
D) sp3d
E) sp3d2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has a nonzero dipole moment?
A) BeCl2
B) SF2
C) KrF2
D) CO2
E) CCl4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 121 - 139 of 139
Related Exams