A) graphite-covalent crystals
B) Na-metallic crystals
C) SiO2-molecular crystals
D) NaCl-ionic crystals
E) diamond-covalent crystals
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Platinum has a face-centered cubic crystal structure and a density of 21.5 g/cm3. What is the radius of a platinum atom?
A) 69 pm
B) 98 pm
C) 139 pm
D) 196 pm
E) 277 pm
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the pair of substances in which the one with the higher vapor pressure at a given temperature is listed first.
A) C7H16, C5H12
B) CCl4, CBr4
C) H2O, H2S
D) CH3CH2OH, CH3-O-CH3
E) Xe, Kr
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Identify the dominant (strongest) type of intermolecular force present in RbCl(s).
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The zincblende structure of ZnS has the relatively large sulfide ions arranged at the lattice points of a face-centered cubic structure. The edge length of this cubic unit cell is 540.9 pm. Determine the density of zincblende.
A) 0.244 g/cm3
B) 1.02 g/cm3
C) 2.05 g/cm3
D) 4.09 g/cm3
E) 8.18 g/cm3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the phase diagram of a pure substance given below, what phase exists at point F? 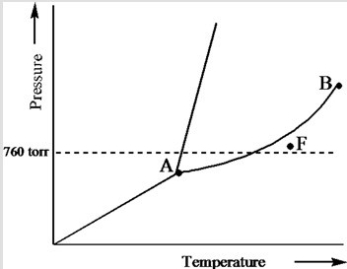
A) vapor + liquid
B) vapor
C) liquid
D) solid
E) supercritical fluid
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Poor conductor of heat and electricity, soft, and low melting point are properties which describe a(n) ________ crystal.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The vapor pressure of ethanol is 400. mmHg at 63.5°C. Its molar heat of vaporization is 39.3 kJ/mol. What is the vapor pressure of ethanol, in mmHg, at 34.9°C? (R = 8.314 J/K • mol)
A) 109 mmHg
B) 220 mmHg
C) 366 mmHg
D) 376 mmHg
E) 1470 mmHg
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Below is the phase diagram for sulfur. Which phase is present at 100°C and 1 atm? 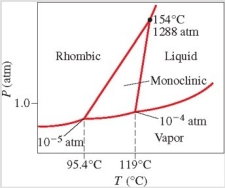
A) liquid
B) vapor
C) rhombic solid
D) monoclinic solid
E) liquid-vapor boundary line
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Octane has a vapor pressure of 40. torr at 45.1°C and 400. torr at 104.0°C. What is its heat of vaporization? (R = 8.314 J/K • mol)
A) 39.1 kJ/mol
B) 46.0 kJ/mol
C) 590 kJ/mol
D) 710 kJ/mol
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Essay
a. Name the two unit cells which occur in close packing of identical atoms. b. Briefly explain how the two types of close-packed lattices of identical atoms differ, in terms of atomic arrangements.
Correct Answer

verified
a. face-centered cubic; hexagonal
b. Cub...View Answer
Show Answer
Correct Answer
verified
b. Cub...
View Answer
Multiple Choice
Which are considered van der Waals forces? 
A) I and III
B) II only
C) III only
D) I, II, and III
E) I only
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following involves ion-dipole interactions?
A) Na+ and Cl-
B) H+ and F-
C) Na+ and Mg2+
D) two water molecules
E) Na+ and H2O
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms does not participate in hydrogen bonding?
A) S
B) O
C) F
D) N
E) H
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following compounds will the molecules not form hydrogen bonds with each other?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Choose the response that lists the member of each of the following pairs that has the higher boiling point. I. H2O or KI, II. HF or HI, III. Cl2 or Br2
A) H2O, HF, and Cl2
B) KI, HF, and Br2
C) KI, HI, and Br2
D) H2O, HI, and Cl2
E) KI, HF, and Cl2
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The surface tension of water is lowered when a detergent is present in solution.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Krypton has a higher melting point than argon because of its
A) hydrogen bonding.
B) stronger dispersion forces.
C) permanent dipole moment.
D) ionic bonds.
E) greater ionization energy.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
At what temperature does ethanol boil on a day in the mountains when the barometric pressure is 547 mmHg? The heat of vaporization of ethanol is 39.3 kJ/mol and its normal boiling point is 78.3°C. (R = 8.314 J/K • mol)
A) 76.5°C
B) 69.9°C
C) 10.0°C
D) 77.9°C
E) 74.6°C
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following factors can contribute to the viscosity for a liquid?
A) Temperature
B) Hydrogen bonding
C) Molecular entanglements
D) London forces
E) All of the answers are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 137
Related Exams