A) I
B) II
C) III
D) IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When forming molecular orbitals from atomic orbitals, what is the order of increasing C-H bond strength for the following set?
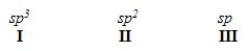
A) II < I < III
B) III < I < II
C) III < II < I
D) I < II < III
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following Lewis structures is correct?
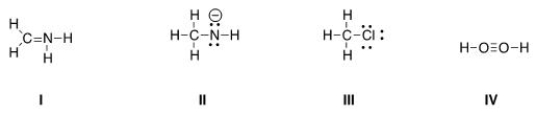
A) I, II
B) I, III
C) II, III
D) III, IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C2H4Cl2?
A) 1
B) 2
C) 3
D) 4
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Avobenzone is an active ingredient in some common sunscreens.Which of the following is the correct molecular formula for avobenzone?
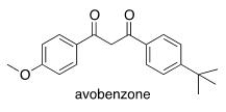
A) C22O22O3
B) C20H22O3
C) C21H23O3
D) C20H24O3
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate H-C-O bond angle in formaldehyde, H2CO?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about bonding is true?
A) Covalent bonds result from the transfer of electrons from one element to another.
B) Ionic bonds result from the transfer of electrons from a metal to a non-metal.
C) Ionic bonds result from the sharing of electrons between two non-metals.
D) Covalent bonds result from the sharing of electrons between two metals.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the approximate C-C-C bond angle in propene, CH3CH = CH2?
A) 90°
B) 109.5°
C) 120°
D) 180°
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has a net dipole moment of zero?

A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a magnesium cation (Mg2+) ?
A) 1s2, 2s2, 2p6
B) 1s2, 2s2, 2p6, 3s1
C) C. 1s2, 2s2, 2p6, 3s2
D) D. 1s2, 2s2, 2p6, 3s2, 3p2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the geometry around the indicated atom in each species. 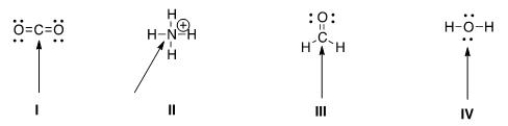
A) I = Linear; II = tetrahedral; III = trigonal planar; IV = tetrahedral
B) I = Linear; II = tetrahedral; III = trigonal planar; IV = linear
C) I = Trigonal planar; II = linear; III = tetrahedral; IV = trigonal planar
D) I = Tetrahedral; II = trigonal planar; III = linear; IV = tetrahedral
F) B) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
Which of the following molecules has polar covalent bonds?
A) MgO
B) NH3
C) Cl2
D) NaBr
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the ground-state electronic configuration of a fluorine atom?
A) 1s2, 2s2, 2p2
B) 1s2, 2s2, 2p3
C) 1s2, 2s2, 2p4
D) 1s2, 2s2, 2p5
F) C) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
In which of the following ions does carbon have a formal charge?
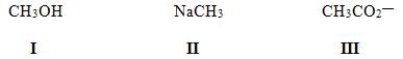
A) I
B) II
C) III
D) None of these
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has nonpolar covalent bonds?
A) HCl
B) N2
C) CHCl3
D) NO
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is the appropriate conversion of (CH3) 2CHOCH2CH2CH2OH to a skeletal structure?
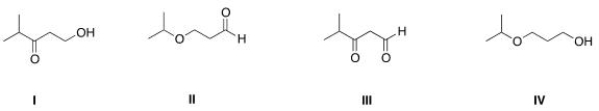
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Arrange the following bonds in decreasing order of ionic character, putting the most ionic first. 
A) I > II > III > IV
B) IV > II > I > III
C) IV > III > II > I
D) IV > II > III > I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following covalent bonds has the largest dipole moment?
A) C-H
B) C-C
C) C-O
D) H-F
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is not true?
A) There is no movement of electrons from one form to another.
B) Resonance structures are not isomers.
C) Resonance structures differ only in the arrangement of electrons.
D) Resonance structures are in equilibrium with each other.
F) C) and D)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
In which structure is the hybridization incorrect?

A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 69
Related Exams