A) 125.9 kJ
B) 252.9 kJ
C) 364.9 kJ
D) 1207.6 kJ
E) None of these choices is correct.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The highly exothermic thermite reaction, in which aluminum reduces iron(III) oxide to elemental iron, has been used by railroad repair crews to weld rails together.  What mass of iron is formed when 725 kJ of heat are released?
What mass of iron is formed when 725 kJ of heat are released?
A) 47 g
B) 65 g
C) 95 g
D) 112 g
E) 130 g
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
In an endothermic reaction, in going from the reactants to the products at the same temperature, the value of q is negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following relationships is always correct?
A) potential energy + kinetic energy = constant
B) E = q + w
C) ( E = H - P V)
D) H = E + PV
E) ( H = qv)
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Use the following data to calculate the standard heat (enthalpy) of formation, Hf, of manganese(IV) oxide, MnO2 (s) . 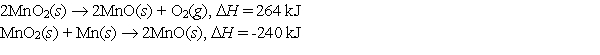
A) -504 kJ
B) -372 kJ
C) -24 kJ
D) 24 kJ
E) 504 kJ
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethanol, C2H5OH, is being promoted as a clean fuel and is used as an additive in many gasoline mixtures. Calculate the H rxn for the combustion of ethanol. ( H f [C2H5OH(l) ] = -277.7 kJ/mol; H f [CO2(g) ] = -393.5 kJ/mol; H f [H2O(g) ] = -241.8 kJ/mol)
A) -1234.7 kJ
B) -751.1 kJ
C) -357.6 kJ
D) 357.6 kJ
E) 1234.7 kJ
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Different chemical bonds have different potential energies.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Natural gas, or methane, is an important fuel. Combustion of one mole of methane releases 802.3 kilojoules of energy. How much energy does that represent in kilocalories?
A) 1.92 *10¯1 kcal
B) 1.92 * 102 kcal
C) 3.36 * 103 kcal
D) 1.92 *105 kcal
E) 3.36 * 106 kcal
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
15.0 g of ice cubes at 0.0 C are combined with 150. g of liquid water at 70.0 C in a coffee cup calorimeter. Calculate the final temperature reached, assuming no heat loss or gain from the surroundings. (Data: specific heat capacity of H2O(l) , c = 4.18 J/g* C; H2O(s) H2O(l) H = 6.02 kJ/mol)
A) 0.0
B) 10.6
C) 30.7
D) 43.2
E) 56.4
G) B) and E)
Correct Answer

verified
Correct Answer
verified
True/False
In a reaction with high energy reactants and low energy products, q is negative.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Stoichiometric amounts of nitrogen gas and hydrogen gas react in a calorimeter to produce 5.00 g of ammonia gas. The calorimeter temperature rises 0.42 C. The calorimeter and water have a combined heat capacity of 32.16 kJ/K. Calculate the heat of formation of ammonia, Hf , in kJ/mol. The formation reaction for ammonia is: 
A) -46 kJ/mol
B) -13.5 kJ/mol
C) -3.97 kJ/mol
D) 3.97 kJ/mol
E) 13.5 kJ/mol
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound carbon suboxide, C3O2, is a gas at room temperature. Use the data supplied to calculate the heat of formation of carbon suboxide. (Data:  H = 127.3 kJ/mol and H f of CO(g) = -110.5 kJ/mol)
H = 127.3 kJ/mol and H f of CO(g) = -110.5 kJ/mol)
A) 116.8
B) -93.7
C) 227.8
D) -348.3
E) 93.7
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A system that does no work but which transfers heat to the surroundings has:
A) q < 0, E > 0
B) q < 0, E < 0
C) q > 0, E > 0
D) q > 0, E < 0
E) q < 0, E = 0
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is a correct formation reaction?
A) C(diamond) C(graphite)
B) H2(g) + O(g) H2O(l)
C) C(graphite) + 4H(g) CH4(g)
D) 6C(graphite) + 6H2O(s) C6H12O6(s)
E) 2C(graphite) + 3H2(g) + ½O2(g) C2H5OH(l)
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a phase change of water between the liquid and the gas phases, 770.1 kJ of energy was released by the system. What was the product, and how much of it was formed in the phase change? (Data: H2O(l) H2O(g) H = 44.01 kJ/mol.)
A) 315 g water vapor was produced.
B) 17.5 g of water vapor was produced.
C) 17.5 mol of water vapor was produced.
D) 17.5 mol of liquid water was produced.
E) 17.5 g of liquid water was produced.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the H rxn for the following reaction. ( H f [AsH3(g) ] = 66.4 kJ/mol; H f [H3AsO4(aq) ] = -904.6 kJ/mol; H f [H2O(l) ] = -285.8 kJ/mol) ![Calculate the \Delta H \degree <sub>rxn</sub> for the following reaction. ( \Delta H \degree <sub>f</sub> [AsH<sub>3</sub>(g) ] = 66.4 kJ/mol; \Delta H \degree <sub>f</sub> [H<sub>3</sub>AsO<sub>4</sub>(aq) ] = -904.6 kJ/mol; \Delta H \degree <sub>f</sub> [H<sub>2</sub>O(l) ] = -285.8 kJ/mol) A) -1981.4 kJ B) -685.2 kJ C) -172.2 kJ D) 172.2 kJ E) 685.2 kJ](https://d2lvgg3v3hfg70.cloudfront.net/TB7799/11eb16b2_ff93_fb2b_984d_e9dcd13fc47f_TB7799_00.jpg)
A) -1981.4 kJ
B) -685.2 kJ
C) -172.2 kJ
D) 172.2 kJ
E) 685.2 kJ
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following is not a correct formation reaction? (products are correct)
A)
B)
C) (graphite)
D) (graphite diamond)
E) graphite
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calcium hydroxide, which reacts with carbon dioxide to form calcium carbonate, was used by the ancient Romans as mortar in stone structures. The reaction for this process is: 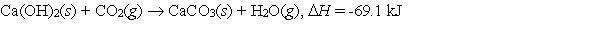 What is the enthalpy change if 3.8 mol of calcium carbonate is formed?
What is the enthalpy change if 3.8 mol of calcium carbonate is formed?
A) -18 kJ
B) -69 kJ
C) -73 kJ
D) -260 kJ
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The only way in which a system can do work on the surroundings is by expansion against the external pressure.
B) False
Correct Answer

verified
Correct Answer
verified
Showing 61 - 79 of 79
Related Exams