A) K
B) Ti
C) Cu
D) Ge
E) Kr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following equations correctly represents the process relating to the ionization energy of X?
A) X(s) X+(g) + e¯
B) X2(g) X+(g) + X¯(g)
C) X(g) + e¯ X¯(g)
D) X¯(g) X(g) + e¯
E) X(g) X+(g) + e¯
G) All of the above
Correct Answer

verified
Correct Answer
verified
True/False
The maximum number of electrons in an atom with the same value of n is 2n2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Select the correct electron configuration for sulfur (Z = 16) .
A) 1s21p62s22p6
B) 1s22s22p83s23p4
C) 1s22s22p83s23p2
D) 1s22s22p63s23p4
E) 1s22s22p63s23d4
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In a single atom, what is the maximum number of electrons that can have quantum number n = 4?
A) 16
B) 18
C) 32
D) 36
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Elements in which the outermost electron has the same principal quantum number n, show similar chemical properties.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the largest second ionization energy (IE2) ?
A) Li
B) B
C) O
D) F
E) Na
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the greatest atomic radius?
A) Li
B) Ne
C) Rb
D) Sr
E) Xe
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
Atomic size decreases across a period due to an increase in the effective nuclear charge, Zeff.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
Define what is meant by electron affinity, and write a balanced chemical equation to represent the relevant process for element Y.
Correct Answer

verified
Electron affinity is the energ...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Hund's rule is used to predict the electron configuration of atoms in excited states.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the element of Period 2 which has the following successive ionization energies, in kJ/mol. 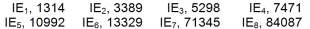
A) Li
B) B
C) O
D) Ne
E) None of these choices is correct.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Elements with ________________ first ionization energies and ___________ electron affinities generally form cations.
A) low, very negative
B) high, positive or slightly negative
C) low, positive or slightly negative
D) high, very negative
E) None of these choices is generally correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
Elements in the same period of the periodic table have similar valence shell electron configurations.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the set of isoelectronic atoms and ions A2¯, B¯, C, D+, and E2+. Which arrangement of relative radii is correct?
A) A2¯ > B¯ > C > D+ > E2+
B) E2+ > D+ > C > B¯ > A2¯
C) A2¯ > B¯ > C < D+ < E2+
D) A2¯ < B¯ < C > D+ > E2+
E) None of these choices is correct.
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following electron configurations is not possible for an atom in an excited state?
A) 1s22s22p63s23p63d104s14p1
B) 1s22s22p63s13p5
C) 1s22s22p63s23p23d2
D) 1s22s22p63s23p63d104s2
E) 1s22s22p63s23p63d104s14p3
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following elements has the smallest atomic size?
A) Na
B) Ar
C) K
D) Ca
E) Kr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following statements about atomic structure and quantum numbers is incorrect?
A) In a given atom, 18 is the maximum number of electrons having principal quantum number n = 3.
B) The number of orbitals in a given f subshell is 7.
C) For n = 4, the largest possible value of l is 3.
D) For n = 4, the largest possible value of ml is 2.
E) The following set of quantum numbers for a single orbital is not allowed: n = 3, l = 1, ml = -2.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
d10np3?
A) 2
B) 3
C) 5
D) 10
E) 15
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
d8?
A) 2
B) 6
C) 8
D) 10
E) None of these choices is correct.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 61 - 80 of 81
Related Exams