A) 520 torr
B) 562 torr
C) 3900 torr
D) 7600 torr
E) 75000 torr
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Mercury is 13.6 times as dense as liquid water. What would be the reading of a water-filled barometer at normal atmospheric pressure, 760. mmHg?
A) 1.03 × 103 torr
B) 1.03 × 103 Pa
C) 1.03 × 103 mm height of water column
D) 1.03 × 103 cm height of water column
E) 13.6 atm
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the root-mean-square speed of methane, CH4 (g) , at 78°C.
A) 23 m/s
B) 350 m/s
C) 550 m/s
D) 667 m/s
E) 740 m/s
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The ozone layer is important because
A) ozone absorbs low energy radiation which warms the troposphere.
B) ozone purifies the atmosphere by reacting with excess fluorocarbons.
C) ozone absorbs ultraviolet radiation.
D) ozone reflects high energy radiation such as X-rays and gamma rays.
E) humans need to breathe air containing some ozone.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following gases effuses most rapidly?
A) Nitrogen
B) Oxygen
C) Hydrogen chloride
D) Ammonia
E) Carbon monoxide
G) B) and E)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
"The total pressure in a mixture of unreacting gases is equal to the sum of the partial pressures of the individual gases" is a statement of __________________ Law.
A) Charles's
B) Graham's
C) Boyle's
D) Avogadro's
E) Dalton's
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 3.0-L sample of helium was placed in a container fitted with a porous membrane. Half of the helium effused through the membrane in 24 hours. A 3.0-L sample of oxygen was placed in an identical container. How many hours will it take for half of the oxygen to effuse through the membrane?
A) 8.5 h
B) 12 h
C) 48 h
D) 60. h
E) 68 h
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 0.750 L of argon at 1.50 atm and 177°C and 0.235 L of sulfur dioxide at 95.0 kPa and 63.0°C are added to a 1.00-L flask and the flask's temperature is adjusted to 25.0°C, what is the resulting pressure in the flask?
A) 0.0851 atm
B) 0.244 atm
C) 0.946 atm
D) 1.74 atm
E) 1.86 atm
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A flask containing argon gas is connected to a closed-ended mercury manometer. The closed end is under vacuum. If the mercury level in the closed arm is 230. mm above that in the arm connected to the flask, what is the argon pressure, in torr?
A) -230.
B) 230.
C) 530.
D) 790.
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 0.850-mole sample of nitrous oxide, a gas used as an anesthetic by dentists, has a volume of 20.46 L at 123°C and 1.35 atm. What would be its volume at 468°C and 1.35 atm?
A) 5.38 L
B) 10.9 L
C) 19.0 L
D) 38.3 L
E) 77.9 L
G) A) and C)
Correct Answer

verified
D
Correct Answer
verified
Multiple Choice
Hydrochloric acid is prepared by bubbling hydrogen chloride gas through water. What is the concentration of a solution prepared by dissolving 225 L of HCl(g) at 37°C and 89.6 kPa in 5.25 L of water?
A) 1.49 M
B) 1.66 M
C) 7.82 M
D) 12.5 M
E) 16.6 M
G) C) and D)
Correct Answer

verified
A
Correct Answer
verified
Multiple Choice
A sample of carbon dioxide gas at 125°C and 248 torr occupies a volume of 275 L. What will the gas pressure be if the volume is increased to 321 L at 125°C?
A) 212 torr
B) 289 torr
C) 356 torr
D) 441 torr
E) 359 torr
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 500-mL sample of argon at 800 torr has its absolute temperature quadrupled. If the volume remains unchanged what is the new pressure?
A) 200 torr
B) 400 torr
C) 800 torr
D) 2400 torr
E) 3200 torr
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Hydrogen peroxide was catalytically decomposed and 75.3 mL of oxygen gas was collected overwater at 25°C and 742 torr. What mass of oxygen was collected? (Pwater = 24 torr at 25°C)
A) 0.00291 g
B) 0.0931 g
C) 0.0962 g
D) 0.0993 g
E) 0.962 g
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the lines on the figure below is the best representation of the relationship between the volume of a gas and its pressure, other factors remaining constant? 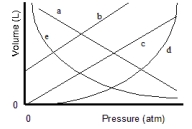
A) a
B) b
C) c
D) d
E) e
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A sample of nitrogen gas is confined to a 14.0 L container at 375 torr and 37.0°C. How many moles of nitrogen are in the container?
A) 0.271 mol
B) 2.27 mol
C) 3.69 mol
D) 206 mol
E) 227 mol
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A carbon dioxide sample weighing 44.0 g occupies 32.68 L at 65°C and 645 torr. What is its volume at STP?
A) 22.4 L
B) 31.1 L
C) 34.3 L
D) 35.2 L
E) 47.7 L
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
According to the kinetic theory of gases, in a collision between two molecules the kinetic energy of one molecule will decrease by the same amount that the kinetic energy of the other one increases.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Calculate the temperature of an argon sample at 55.4 kPa and 18.6 L if it occupies 25.8 L at 75.0°C and 41.1 kPa.
A) 95.0°C
B) 85.1°C
C) 77.2°C
D) 72.9°C
E) 65.2°C
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If the molecular mass of a gas increases by a factor of 4 at constant temperature, its rms speed will
A) decrease by a factor of 4.
B) increase by a factor of 4.
C) decrease by a factor of 16.
D) increase by a factor of 16.
E) decrease by a factor of 2.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 97
Related Exams