A) 1s2,2s2,2p6
B) 1s2,2s2,2p6,3s2,3p6
C) 1s2,2s2,2p6,3s2,3p5
D) 1s2,2s2,2p6,3s2,3p4
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about constitutional isomers is true?
A) Constitutional isomers are different molecules having the different molecular formula.
B) Constitutional isomers are different molecules having the same molecular formula.
C) Constitutional isomers are same molecules having the different molecular formula.
D) Constitutional isomers are same molecules having the same molecular formula.
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following atoms in order of decreasing electronegativity,putting the most electronegative first. 
A) I > IV > II > III
B) II > III > IV > I
C) III > IV > II > I
D) III > II > IV > I
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds are non-polar? 
A) I,IV
B) I,II
C) II,III
D) II,IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is not true?
A) There is no movement of electrons from one form to another.
B) Resonance structures are not isomers.
C) Resonance structures differ only in the arrangement of electrons.
D) Resonance structures are in equilibrium with each other.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Convert the following skeletal structure to a condensed structure. 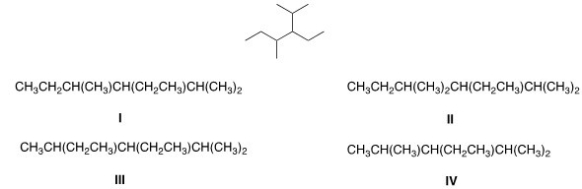
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Rank the following in order of decreasing importance as contributing structures to the resonance hybrid of formaldehyde,H2CO. 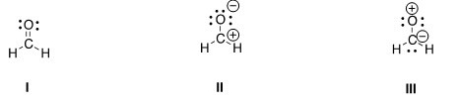
A) I > II > III
B) I > III > II
C) II > I > III
D) III > II > I
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many different isomers are there for a compound having the molecular formula C3H6O?
A) 4
B) 5
C) 6
D) 7
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would most likely form an ionic bond? 
A) I
B) II
C) III
D) IV
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has nonpolar covalent bonds?
A) HCl
B) N2
C) CHCl3
D) NO
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization of the carbon atom in the methyl cation,(CH3+) ?
A) sp3
B) sp2
C) sp
D) p
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular geometry around the carbon atom in CH4?
A) Tetrahedral
B) Trigonal Planar
C) Trigonal Pyramidal
D) Linear
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the order of decreasing bond length for a C-C bond composed of the following molecular orbitals? 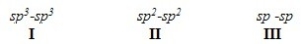
A) I > III > II
B) I > II > III
C) III > II > I
D) II > III > I
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many constitutional isomers are there for a molecule having the molecular formula C2H4Cl2?
A) 1
B) 2
C) 3
D) 4
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic orbitals overlap to form the carbon-carbon s and p bonding molecular orbitals of ethylene,H2C=CH2?
A) Csp3 + Csp3,and C2p + C2p
B) Csp3 + Csp3,and Csp2 + Csp2
C) Csp2 + Csp2,and C2p + C2p
D) Csp2 + Csp2,and Csp2 + Csp2
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules has polar covalent bonds?
A) MgO
B) NH3
C) Cl2
D) NaBr
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about valence electrons is true?
A) They are the most tightly held electrons.
B) They do not participate in chemical reactions.
C) They are the outermost electrons.
D) They reveal the period number of a second-row element.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following atoms will have a full 3s orbital in the ground state?
A) Hydrogen
B) Lithium
C) Potassium
D) Rubidium
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct Lewis structure for acetic acid (CH3CO2H) ? 
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following molecules contain both covalent and ionic bonds? 
A) I,II
B) I,IV
C) II,III
D) II,IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 60 of 77
Related Exams