A) One s bond and one p bond.
B) One s bond and two p bonds.
C) Two s bonds and one p bond.
D) Two s bonds and two p bonds.
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 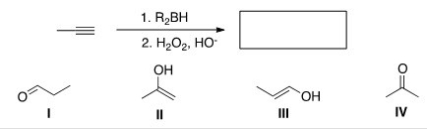
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following explains why 1-pentyne has a slightly higher boiling point than 1-pentene?
A) 1-Pentyne has more carbons than 1-pentene.
B) 1-Pentyne has more carbons per hydrogen than 1-pentene.
C) 1-Pentyne is linear while 1-pentene is trigonal planar.
D) The C-C triple bond in 1-pentyne is more polar than the C-C double bond in 1-pentene.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 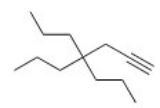
A) 4,4-Diethyl-1-octyne
B) 4,4-Dipropyl-1-heptyne
C) 4,4-Dipropyl-6-heptyne
D) 4,4-Diethyl-6-octyne
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the IUPAC name for the following compound. 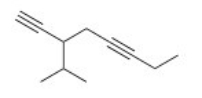
A) 3-sec-Butyl-1,4-octadiyne
B) 3-Isopropyl-1,5-octadiyne
C) 3-Isopropyl-1,5-nonadiyne
D) 3-sec-Butyl-1,4-nonadiyne
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the starting material in the following reaction? 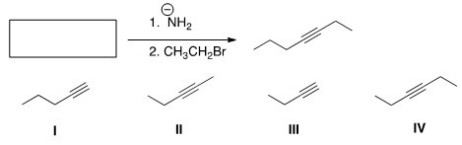
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following bases cannot deprotonate acetylene? You are given the pKa values of the conjugate acids in parentheses. 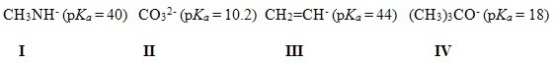
A) Only I and II
B) Only I and III
C) Only II and III
D) Only II and IV
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the product of the following reaction. 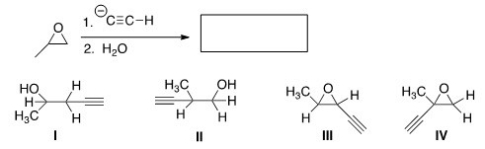
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about alkynes is not true?
A) Alkynes undergo many addition reactions.
B) Alkynes are more polarizable than alkenes.
C) Both p bonds of a C-C triple bond are weaker than a C-C s bond.
D) The electrons in the p bonds of alkynes are more tightly held than those of alkenes.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 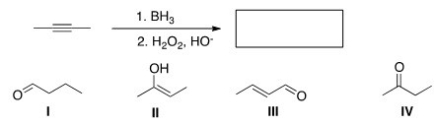
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the indicated hydrogen in the following compounds is the most acidic? 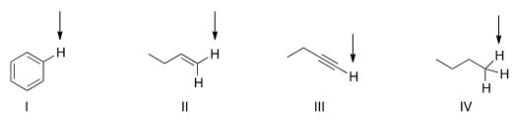
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 51 of 51
Related Exams