A) Three π bonds break; two σ bonds and one π bond form.
B) Three π bonds break; one σ bond and two π bonds form.
C) Two π bonds break; one σ bond and one π bond form.
D) Two π bonds break; two σ bonds and one π bond form.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the major product(s) of the following reaction? 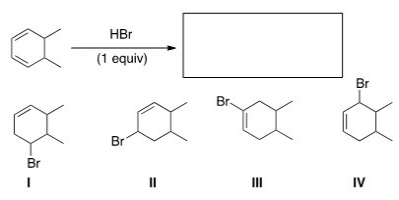
A) Only I
B) Only II and IV
C) Only I and III
D) Only I,II,and III
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is conjugated? 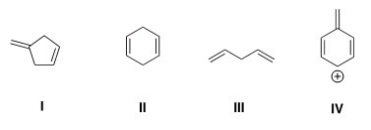
A) I
B) II
C) III
D) IV
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 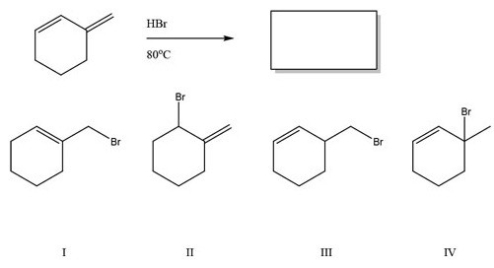
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the definition of a resonance structure?
A) Compounds with the same molecular formula
B) Compounds with different carbon frameworks
C) Structures that differ only in the placement of π and nonbonding electrons
D) Structures that differ only in the location of bonds
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 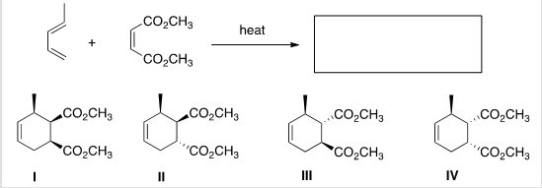
A) I
B) II
C) III
D) IV
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization around the indicated oxygen atom in the following anion? 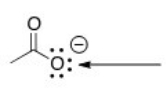
A) sp3
B) sp2
C) sp
D) p
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 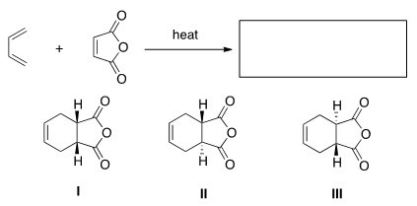
A) Only I
B) Only I and II
C) Only II and III
D) I,II,and III
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major organic product of the following reaction? 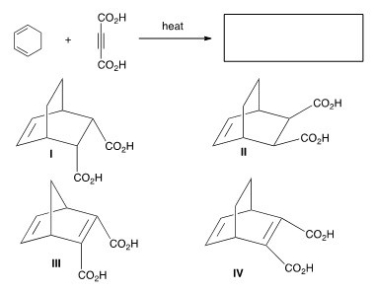
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is the least reactive dienophile in a Diels-Alder reaction? 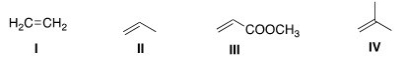
A) I
B) II
C) III
D) IV
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How would you favor the formation of the kinetic product of the following reaction? 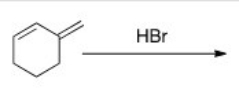
A) Excess HBr
B) Long reaction time
C) Low temperature
D) High pressure
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a resonance structure of acetic anhydride? 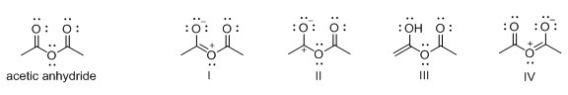
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is (are) the major product(s) of the following reaction? 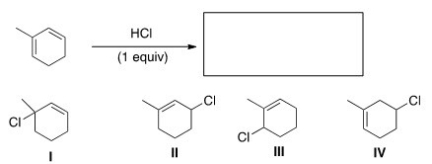
A) Only I
B) Only I and III
C) Only II and IV
D) Only I,II,and III
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why would the compound below not react with a dienophile in a Diels-Alder reaction? 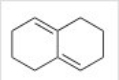
A) The compound is not a conjugated diene.
B) There are no electron withdrawing groups on the compound.
C) There are no electron donating groups on the compound.
D) The compound cannot adopt the s-cis conformation.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which triene has the smallest heat of hydrogenation? 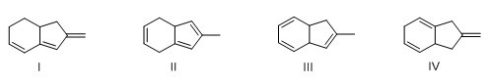
A) I
B) II
C) III
D) IV
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the hybridization around the indicated carbon atom in the following anion? 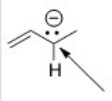
A) sp3
B) sp2
C) sp
D) p
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compound absorbs UV light at the longest wavelength? 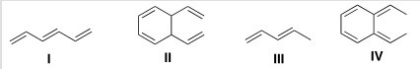
A) I
B) II
C) III
D) IV
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following conjugated dienes represent two conformations? 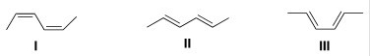
A) Only I and II
B) Only I and III
C) Only II and III
D) None of the choices
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about resonance structures is true?
A) The placement of atoms is different.
B) The placement of p bonds is different.
C) The placement of s bonds is different.
D) The placement of nonbonded electrons is the same.
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the major product of the following reaction? 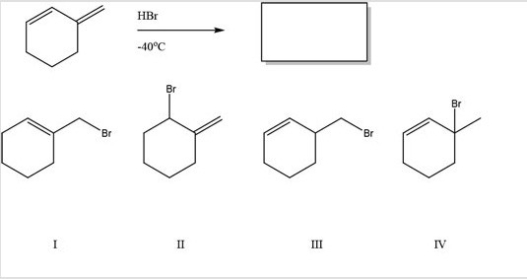
A) I
B) II
C) III
D) IV
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 53
Related Exams