A) Keq > 1 when DG° > 0
B) Keq > 1 when DG° < 0
C) Keq < 1 when DG° < 0
D) Keq < 1 when DG° = 0
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: 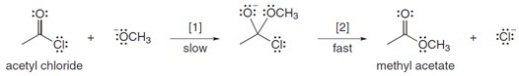 The conversion of acetyl chloride to methyl acetate would be classified as which of the following?
The conversion of acetyl chloride to methyl acetate would be classified as which of the following?
A) Addition
B) Elimination
C) Substitution
D) None of these
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which reaction is slowest? 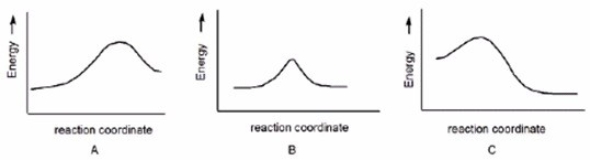
A) A
B) B
C) C
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true?
A) The product is favored in reaction in which DH° is a positive value.
B) Entropy decreases when an acyclic compound forms a ring.
C) In homolytic bond cleavage,entropy decreases and favors formation of products.
D) The starting material is favored in a reaction in which DH° is a negative value.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What kind of reaction does the conversion of A to D represent? 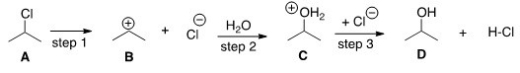
A) Addition reaction
B) Substitution reaction
C) Elimination reaction
D) Oxidation-reduction reaction
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The symbol hν stands for ________ in a chemical reaction.
A) light
B) heat
C) reactant
D) product
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is true?
A) Bond dissociation energies increase down a column of the periodic table.
B) When DH° is positive,more energy is released in forming bonds than is needed to break bonds.
C) When DH° is negative,more energy is needed to break bonds than is released in forming bonds.
D) Bond dissociation energies decrease down a column of the periodic table.
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about a two-step reaction mechanism is true?
A) The transition states are located at energy minima.
B) Each step is characterized by its own value of DH° and Ea.
C) The rate-determining step has the lower energy transition state.
D) The reactive intermediate is located at an energy maximum.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many transition states are present in the reaction in the energy diagram? 
A) 0
B) 1
C) 2
D) 3
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What kind of reaction does the conversion of A to B represent? 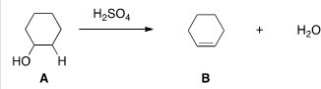
A) Addition reaction
B) Elimination reaction
C) Substitution reaction
D) Oxidation-reduction reaction
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: ![The conversion of acetyl chloride to methyl acetate occurs via the following two-step mechanism: What is the rate equation for this reaction if the first step is rate determining? A) Rate = k [acetyl chloride] [<sup>-</sup>OCH<sub>3</sub>] B) Rate = k [acetyl chloride] C) Rate = k [<sup>-</sup>OCH<sub>3</sub>] D) Rate = k [acetyl chloride] [<sup>-</sup>OCH<sub>3</sub>]<sup>2</sup>](https://d2lvgg3v3hfg70.cloudfront.net/TB7662/11eac43c_f7f5_469b_a9af_3d93d8b1c97e_TB7662_00.jpg) What is the rate equation for this reaction if the first step is rate determining?
What is the rate equation for this reaction if the first step is rate determining?
A) Rate = k [acetyl chloride] [-OCH3]
B) Rate = k [acetyl chloride]
C) Rate = k [-OCH3]
D) Rate = k [acetyl chloride] [-OCH3]2
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which reaction is Keq > 1? 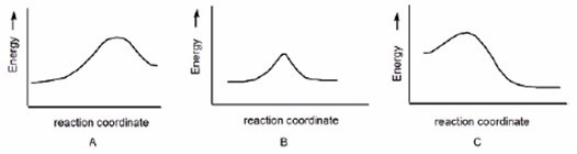
A) A
B) B
C) C
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which step would most likely have the largest energy of activation? 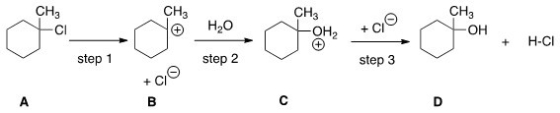
A) Step one
B) Step two
C) Step three
D) It cannot be determined from the information provided
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 41 - 53 of 53
Related Exams